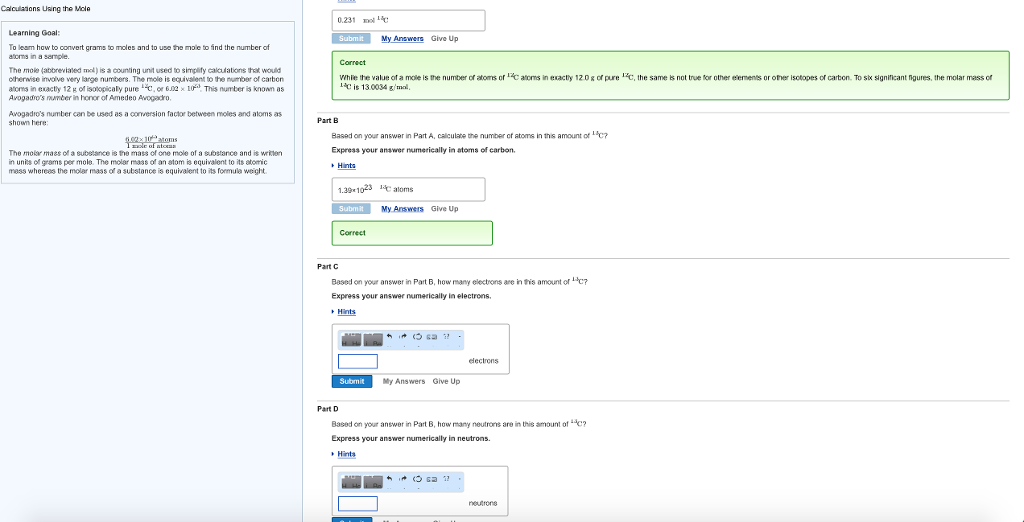
Calculations Using the Mole Learning Goal: To learn how to convert grams to moles and to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Calculations Using the Mole Learning Goal: To learn how to convert grams to moles and to use the mole to find the number of ators in a sample. The mole (abbreviated mol) is a counting unit used to simplify calculations that would otherwise involve very large numbers. The mole is equivalent to the number of carbon atoms in exactly 12g of isotopically pure *C, or 6.02 x 10 This number is known as Avogadro's number in honor of Amedeo Avogadro. Avogadro's number can be used as a conversion factor between moles and atoms as shown here: 40210 atoms 1 mole of atomas The molar mass of a substance is the mass of one mole of a substance and is written in units of grams per mole. The molar mass of an atom is equivalent to its atomic mass whereas the molar mass of a substance is equivalent to its formula weight. 0.231 mol c Submit My Answers Give Up Correct While the value of a mole is the number of atoms of C atoms in exactly 12.0 g of pure C, the same is not true for other elements or other isotopes of carbon. To six significant figures, the molar mass of acis 13.0034 g/mol. Part B Based on your answer in Part A, calculate the number of atoms in this amount of C? Express your answer numerically in atoms of carbon. Hints 1.39×1023 Caloms Submit My Answers Give Up Correct Part C Based on your answer in Part B, how many electrons are in this amount of c Express your answer numerically in electrons. Submit (Osa 17- My Answers Give Up electrons Part D Based on your answer in Part B, how many neutrons are in this amount of **C? Express your answer numerically in neutrons. Hints (Osa 17. neutrone Calculations Using the Mole Learning Goal: To learn how to convert grams to moles and to use the mole to find the number of ators in a sample. The mole (abbreviated mol) is a counting unit used to simplify calculations that would otherwise involve very large numbers. The mole is equivalent to the number of carbon atoms in exactly 12g of isotopically pure *C, or 6.02 x 10 This number is known as Avogadro's number in honor of Amedeo Avogadro. Avogadro's number can be used as a conversion factor between moles and atoms as shown here: 40210 atoms 1 mole of atomas The molar mass of a substance is the mass of one mole of a substance and is written in units of grams per mole. The molar mass of an atom is equivalent to its atomic mass whereas the molar mass of a substance is equivalent to its formula weight. 0.231 mol c Submit My Answers Give Up Correct While the value of a mole is the number of atoms of C atoms in exactly 12.0 g of pure C, the same is not true for other elements or other isotopes of carbon. To six significant figures, the molar mass of acis 13.0034 g/mol. Part B Based on your answer in Part A, calculate the number of atoms in this amount of C? Express your answer numerically in atoms of carbon. Hints 1.39×1023 Caloms Submit My Answers Give Up Correct Part C Based on your answer in Part B, how many electrons are in this amount of c Express your answer numerically in electrons. Submit (Osa 17- My Answers Give Up electrons Part D Based on your answer in Part B, how many neutrons are in this amount of **C? Express your answer numerically in neutrons. Hints (Osa 17. neutrone
Expert Answer:
Answer rating: 100% (QA)
Answer Part 6 In 1 The Part D one electrons atom of 139 x 102... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Use Exercise 68 to find the number of different equivalence relations on a set with n elements, where n is a positive integer not exceeding 10. Let p(n) denote the number of different equivalence...
-
Use Chebyshev's inequality to find the number of times we need to flip an unbiased coin to be able to assert with a probability of at most 0.01 that the proportion of tails will be at most 0.04.
-
Use numbers 1 to 5 to identify each carbon atom in the product of this reaction. What coenzyme is involved? "OOC-CH2-CH2- COO--OOC-CH NH CH NH.
-
. The following is given: EBIT = $50 000, Q=60 000, P =$6, V = $4. a) If sales should increase by 10%, by what percent would EBIT increase? b) If sales should increase by 10%, for what interest...
-
Hyacinth Macaw invests 60% of her funds in stock I and the balance in stock J. The standard deviation of returns on I is 10%, and on J it is 20%. Calculate the variance of portfolio returns, assuming...
-
If two electrons are each 1.50 x 10 -10 m from a proton, as shown in Fig. E 21.37, find the magnitude and direction of the net electric force they will exert on the proton. Figure E21.37 65.0
-
Fossil is famous for fashion wristwatchcs and leather goods. At the end of a recent year. Fossils total assets added up to \($345\) million, and stockholders equity was \($240\) million. How much...
-
1. What is the purpose of a cash flow budget? What does it reveal? Of what value would it be to Regina? 2. How does the payback method work? How does the net present value method work? How would you...
-
Financial statement data for the years ending December 31, 20Y3 and 20Y2, for Lawson Company follow: Sales Total assets: Beginning of year End of year 20Y3 $1,400,000 610,000 790,000 20Y2 $1,026,000...
-
The Garden Ornament Company manufactures two types of garden ornament: a duck and a heron. The information presented in Tables T 1 to T 5 has been prepared, as a result of discussions by line...
-
Champion Rent-All, a business in Brandon Manitoba, rents out tools and equipment. The accounts for the business are as follows: Bank AP - various AR-various Supplies Rental Tools and Equipment Truck...
-
Step 1: In this assignment, you will modify the student information program described above in the module note "Work with Classes and Files." Before we start, please make sure you have followed the...
-
Read the scenario and use the provided information to complete the assignment. Scenario During the current tax year, William and Mary had various stock transactions that qualified as capital gains....
-
Magic Realm, Incorporated, developed a new fantasy board game and sold 5 9 , 4 0 0 units last year at a selling price of $ 6 8 per game. Fixed expenses associated with the game are $ 1 , 0 8 9 , 0 0...
-
Table 4.8 Level, Slope and Curvature 3 month 6 month 1 year 2 year 7 year 10 year 1.0111 1.0180 ' (Level) 1.0344 1.0299 -0.3507 -0.1424 1.0180 0.2432 0.5205 0.7432 1.0180 0.9509 0.9196 (Slope)...
-
a) Determine the differential equation that relates the input and output of the system. b) Find the impulse response of the system. c) Find the output of the system for input x(t) = eu(t)(1-t)...
-
Write script to do the following: Use create table statement to create new copy of the L_EMPLOYEES table, with a new name. Then use an insert statement with a select clause to copy all the data from...
-
$10,000 was borrowed at 3.5% on July 17. The borrower repaid $5000 on August 12, and $2000 on September 18. What final payment is required on November 12 to fully repay the loan?
-
At 303 K, the vapor pressure of benzene is 120. Torr and that of hexane is 189 Torr. Calculate the vapor pressure of a solution for which x benzene = 0.28 assuming ideal behavior.
-
Molecules such as dimethylsulfoxide and dimethylsulfone can either be represented as hypervalent, that is, with more than the normal complement of eight valence electrons around sulfur, or as...
-
How can circular dichroism spectroscopy be used to determine the secondary structure of a bio-molecule?
-
What is a best practice in general? Give examples of best practices in an area unrelated to project management, such as nutrition, exercise, or child rearing.
-
What are some unique features of project management software in general?
-
Why should organizations identify and use best practices? What are the main categories of best practices developed as part of OPM3?

Study smarter with the SolutionInn App


