Calculate the pH of the following solution. All concentrations are 0.0100 M. The k of Acetic...
Fantastic news! We've Found the answer you've been seeking!
Question:
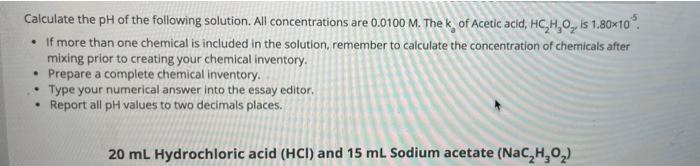
Transcribed Image Text:
Calculate the pH of the following solution. All concentrations are 0.0100 M. The k of Acetic acid, HC,H,O, is 1.80x10. • If more than one chemical is included in the solution, remember to calculate the concentration of chemicals after mixing prior to creating your chemical inventory. • Prepare a complete chemical inventory. Type your numerical answer into the essay editor. Report all pH values to two decimals places. 20 ml Hydrochloric acid (HCI) and 15 mL Sodium acetate (Nac,H,0,) Calculate the pH of the following solution. All concentrations are 0.0100 M. The k of Acetic acid, HC,H,O, is 1.80x10. • If more than one chemical is included in the solution, remember to calculate the concentration of chemicals after mixing prior to creating your chemical inventory. • Prepare a complete chemical inventory. Type your numerical answer into the essay editor. Report all pH values to two decimals places. 20 ml Hydrochloric acid (HCI) and 15 mL Sodium acetate (Nac,H,0,)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the pH of the following two buffer solutions: (a) 2.0 M CH3COONa/2.0 M CH3COOH, (b) 0.20 M CH3COONa/0.20 M CH3COOH. Which is the more effective buffer? Why?
-
Calculate the pH of a solution made up from 2.0 g of potassium hydroxide dissolved in 115 mL of 0.19 M perchloric acid. Assume the change in volume due to adding potassium hydroxide is negligible.
-
Calculate the pH of a solution made by adding 2.50 g of lithium oxide (Li2O) to enough water to make 1.500 L of solution.
-
Required A. Calculate Mr. Worthy's minimum employment income for the current year. B. Assume Mr. Worthy had only $4,000 in commission income in addition to his $65,000 salary. Calculate Mr. Worthy's...
-
Hair care companies everywhere spend a bundle on ads in trendy fashion magazines and in television advertising campaigns. But one niche player, Bumble and bumble (B&b), has a different strategythey...
-
Atlantic Resource Management, LLC, to collect a debt from Michael E. Engler. Atlantic called Engler's employer and asked his supervisor about the company's policy concerning the execution of...
-
Let us consider three consecutive flips of a fair and memoryless coin. There are two outcomes H and T for each flip, and eight possible scenarios: we deal with an algebra including events W = (H, H,...
-
In a material processing experiment conducted aboard the space shuttle, a coated niobium sphere of l0-mm diameter is removed from a furnace at 900C and cooled to a temperature of 300C. Although...
-
Suppose a doctors order instructs the infusion of 240mL of a certain medicine over 60 minutes with a drip factor of 20 gtt/mL. (Note: gtt/mL means drops per milliliter.) Determine how many drops per...
-
At April 30, partners capital balances in YBG Company are: Younger $30,000, Beyer $16,000, and Giger $10,000. The income-sharing ratios are 5:3:2, respectively. On May 1, the YBGE Company is formed...
-
Compare the Euler crippling loads of two columns-one of solid circular section and the second of hollow circular section of internal diameter 70% of the external diameter, if they are of the same...
-
Capital Structure Debt 40% Interest rate 5% Tax Rate 26% Equity 60% Risk Free rate 6% RM 13% Beta 1% Working capital 10% next year's sales No terminal cash flows Project 1 Capital investment...
-
As of January 2020, the average pay for a Level 1 Registered Nurse (RN) is $63,190 annually. Leo is a registered nurse (RN) with gross earnings of $2,334.04 semimonthly. If the hospital where Leo...
-
Explore the relevance of the text to you and others: personal connection, other texts, other people What is the effect of this image on you as the viewer, and/or how might it affect other viewers?...
-
Image transcription text How do remarkable checks and stores on the way influence the bank compromise process? 6. What are a few normal reasons for inconsistencies between the bank proclamation and...
-
Research should be taken from theory into practice. Consider what you have learned so far in this course about different research methods and leadership. What can you utilize professionally right...
-
Barrett's WACC is 12%, the market value of its debt and preferred stock totals $60 million, and it has 10 million shares of common stock outstanding. a. What is the present value of the free cash...
-
Eleni Cabinet Company sold 2,200 cabinets during 2011 at $160 per cabinet. Its beginning inventory on January 1 was 130 cabinets at $56. Purchases made during the year were as follows: February . 225...
-
Commercially, compressed oxygen is sold in metal cylinders. If a 120-L cylinder is filled with oxygen to a pressure of 132 atm at 22C, what is the mass (in grams) of O2 present? How many liters of O2...
-
When 2.50 g of a zinc strip were placed in a AgNO3 solution, silver metal formed on the surface of the strip. After some time had passed, the strip was removed from the solution, dried, and weighed....
-
Which member of each of the following pairs of substances would you expect to have a higher boiling point? (a) O2 and Cl2, (b) SO2 and CO2, (c) HF and HI
-
Classify each of the following as either a policy instrument or an intermediary target. Explain your answer. a. Long-term interest rates b. Central bank interest rates c. M2 d. Reserve requirements
-
The New York Fed executes foreign exchange interventions for the Federal Reserve system. Go to https://www.newyorkfed.org/markets/quar_reports.html to see quarterly summaries of the Feds foreign...
-
Calculate the overvaluation of the Thai baht (THB) if you can get 34.6 THB per USD at the exchange counter, but a lunch menu that costs 25 USD in Boston sells for 948.25 THB in Bangkok.

Study smarter with the SolutionInn App


