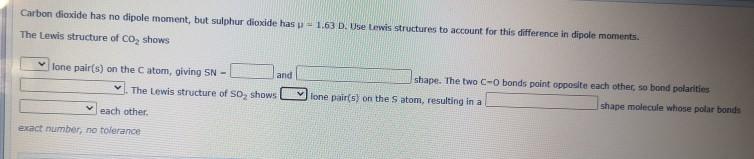
Carbon diaxide has no dipole moment, but sulphur dioxide has p 1.63 D. Use Lewis structures...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Carbon diaxide has no dipole moment, but sulphur dioxide has p 1.63 D. Use Lewis structures to account for this difference in dipole moments. The Lewis structure of CO, shows lone pair(s) on the C atom, giving SN - shape. The two C-0 bonds paint opposite each other, so bond polarities and The Lewis structure of So, shows V lone pair(s) on the S atom, resulting in a shape molecule whose polar bonds each other. exact number, no tolerance Carbon diaxide has no dipole moment, but sulphur dioxide has p 1.63 D. Use Lewis structures to account for this difference in dipole moments. The Lewis structure of CO, shows lone pair(s) on the C atom, giving SN - shape. The two C-0 bonds paint opposite each other, so bond polarities and The Lewis structure of So, shows V lone pair(s) on the S atom, resulting in a shape molecule whose polar bonds each other. exact number, no tolerance
Expert Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these chemistry questions
-
Boron trifluoride (BF3) has no dipole moment ( = 0 D). Explain how this observation confirms the geometry of BF3 predicted by VSEPR theory.
-
A molecule XF6 (having no lone pairs) has a dipole moment of zero. (X denotes an unidentified element.) When two atoms of fluorine have been taken away, you get the molecule XF4, which has dipole...
-
The ammonia molecule (NH3) has a dipole moment of 5.0 X 10-30 C m. Ammonia molecules in the gas phase are placed in a uniform electric field E with magnitude 1.6 X 106 N/c. (a) What is the change in...
-
PacTec Luggage Shop is a small retail establishment located in a large shopping mall. This shop has implemented the following procedures regarding inventory items: a. Since the display area of the...
-
Describe the three factors that can make a t value larger.
-
Write the half-cell reactions and the balanced chemical equation for the electrochemical cells diagrammed here. Use data from Table 19.1 and Appendix D to calculate E cell for each reaction. Table...
-
Determine algebraic equations for the pump head rise, power, and efficiency as functions of flow rate based on the data from Problem 12.18. Analytically determine the Best Efficiency Point and the...
-
Starwood Hotels & Resorts Worldwide provides lodging services around the world. The company is separated into two major divisions. Hotel Ownership: Hotels owned and operated by Starwood. Vacation...
-
Suppose a point charge creates a 1 2 5 0 0 N / C electric field at a distance of 0 . 3 5 m . What is the magnitude of the point charge in coulombs? What is the strength of the field, in newtons per...
-
Based on the characteristics below and creating a proforma and 10 year Discounted cash Flow analysis what would the answers to the questions below be. 1. Assume to cost of capital for the equity...
-
Find the equations of three level curves of the function f (x, y) = e* + cosy. Your answer should show the equation, direction, and location of each of these curves. The three curves should be in...
-
You are an investment banker in a major US firm and receive info from time to time from corporate clients re: takeover activities. A friend who is a securities trader in another brokerage firm has...
-
1 Write a program that calculates and prints the average of several integers. Assume the last value read with scanf is the sentinel 9999. An example input sequence would be: 10 8 11 7 9 9999...
-
Last year, Industrial Industries had Operating Cash Flow of 235,000. It had Net Capital Spending of 445,000 and an investment in Net Working Capital of 155,000. What was its Cash Flow from Assets (or...
-
Name and explain 2 differences between using oil futures to fix the price of future purchases and doing the same using swaps.
-
Consider a 30-year U.S. corporate bond paying 3.5 percent coupon. The bond has 17 years left to maturity andis currently priced at $980. The bond is callable in 8 years at a 6 percent call premium....
-
As you create a budget, try to think about your life ten years from now. Each instruction below will guide you step by step. You've need to do some research on the internet. For example, if you...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
Determine the second-law efficiency of the compression process described in Prob. 12-98E. Take T0 = 77oF.
-
The pressures just upstream and downstream of a hydraulic turbine are measured to be 1325 kPa and 100 kPa, respectively. What is the maximum work, in kJ/kg, that can be produced by this turbine? If...
-
Determine the COP of a refrigerator that removes heat from the food compartment at a rate of 5040 kJ/h for each kW of power it consumes. Also, determine the rate of heat rejection to the outside air.
-
A university is deciding between two meal plans. One plan charges a fixed fee of \($600\) per semester and allows students to eat as much as they want. The other plan charges a fee based on the...
-
Evaluate this statement: You are a natural athlete, an attractive person who learns easily and communicates well. Clearly, you can do everything better than your friends and acquaintances. As a...
-
In elementary school and through middle school, most students have the same teacher throughout the day and for the entire school year. Then, beginning in high school, different subjects are taught by...

Study smarter with the SolutionInn App



