Question: Chapter 16 Problem 26 Par A Which of the following reactions can be used to prepare 3-methyl-3-hexanol, OH CH3CH2CCH2CH2CH3 ? CH 3 Choose all
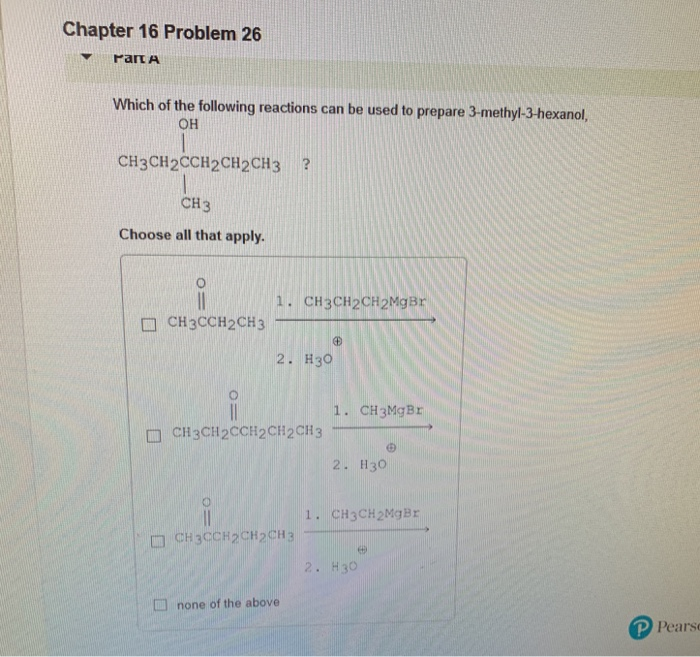
Chapter 16 Problem 26 Par A Which of the following reactions can be used to prepare 3-methyl-3-hexanol, OH CH3CH2CCH2CH2CH3 ? CH 3 Choose all that apply. 0 CH3CCH2CH3 1. CH3CH2CH2MgBr 2. H30 CH3CH2CCH2CH2CH3 CH3CCH2CH2CH3 none of the above 1. CH 3MgBr 2. H30 1. CH3CH2MgBr 2. H30 6 P Pearse
Step by Step Solution
3.41 Rating (151 Votes )
There are 3 Steps involved in it
Ans would ... View full answer

Get step-by-step solutions from verified subject matter experts


