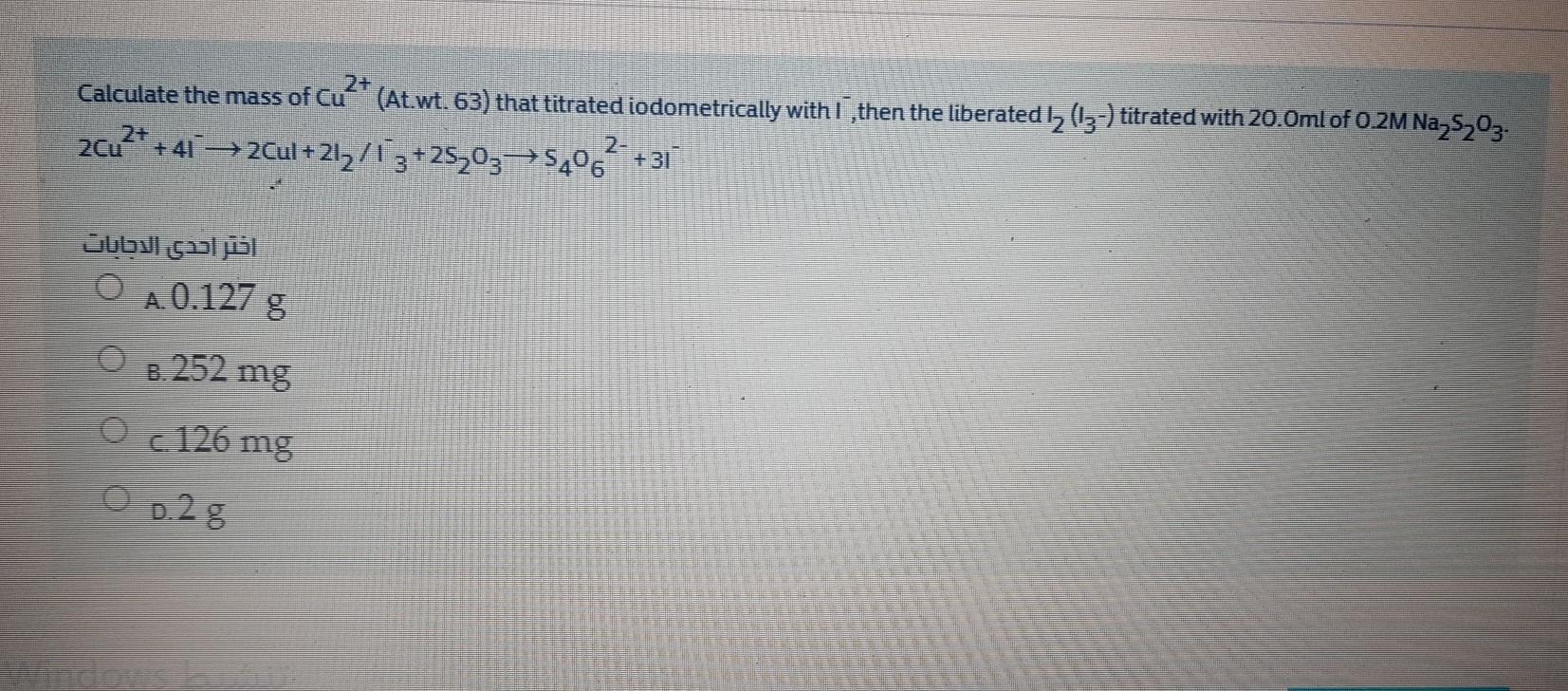
2+ Calculate the mass of Cu (At.wt. 63) that titrated iodometrically with I ,then the liberated...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2+ Calculate the mass of Cu (At.wt. 63) that titrated iodometrically with I ,then the liberated I, (-) titrated with 20.0ml of 0.2M Na,S,0. 2- +41 2Cul + 21, / 13+25,03→$,06 +31 Jubul çal jisl A.0.127 g B. 252 mg Oc 126 mg O D.2 8 ndows 2+ Calculate the mass of Cu (At.wt. 63) that titrated iodometrically with I ,then the liberated I, (-) titrated with 20.0ml of 0.2M Na,S,0. 2- +41 2Cul + 21, / 13+25,03→$,06 +31 Jubul çal jisl A.0.127 g B. 252 mg Oc 126 mg O D.2 8 ndows
Expert Answer:
Answer rating: 100% (QA)
The given question is from iodometric titration This titration is generally used for determining the ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the mass of an atom of (a) helium, (b) iron, and (c) lead. Give your answers in grams. The atomic masses of these atoms are 4.00 u, 55.9 u, and 207 u, respectively.
-
Calculate the mass of the deuteron given that the first line in the Lyman series of H lies at 82259.098 cm-1 whereas that of D lies at 82 281.476 cm-1 Calculate the ratio of the ionization energies...
-
Calculate the mass of the precipitate formed when 2.27 L of 0.0820 M Ba(OH)2 are mixed with 3.06 L of 0.0664 M Na2SO4.
-
Blaine is a practicing dentist. He operates his business from the basement of his house, with a separate entrance and facilities. Blaine uses the simplified method every year to calculate his...
-
What is a prejudgment attachment? What is a writ of execution? How does a creditor use these remedies?
-
A university registrars office maintains data about the following entities: (a) Courses, including number, title, credits, syllabus, and prerequisites; (b) Course offerings, including course number,...
-
Cottle Ltd uses a sales journal, a cash receipts journal, a general journal and an accounts receivable subsidiary ledger within a periodic inventory system. The terms of all credit sales are 2/10,...
-
Contingent liabilities present certain problems and opportunities for companies. You have been asked to help prepare the companys annual report and a lawsuit has been brought against the company for...
-
1. 2. (c) Be(OH)2 (d) Sr(OH)2 Number of amphoteric compounds among the following is (a) BeO (B) Bao The stepwise formation of [Cu(NH)]* is given below: C ANH, LE [Cu(NH, ) [Cu(NH)]+NH, [Cu(NH3)2]+NH,...
-
You are asked to evaluate employees' exposures to methyl n-amyl ketone during a painting operation using NIOSH Method 2553 for the sampling. You can access the method by clicking the link below: ...
-
Financial Planning al Biriktir just graduated with a B . S . degree in engineering and landed a new job with a starting annual gross salary of $A . There are a number of things that he would like to...
-
6. Payments of 300, 500 and 700 are made at the end of years five, six and eight, respectively. Interest is accumulated at an annual effective rate of 4%. You are to find the point in time at which a...
-
1 . Why is scientific rigor important? How is the rigor for qualitative research different than quantitative? 2 . Describe phenomenological, grounded theory, ethnography, and exploratory research...
-
Interpret the results below in both a time-series and cross-sectional manner (by separating the operating drivers of ROE from the financial drivers of ROE). RATIO ANALYSIS LOCKHEED 2021 Profitability...
-
For your company, assuming an anticipated 10% sales increase in the following year calculate the following: a. Working Assets in the most recent year that you have data for. b. Working Liabilities in...
-
Mr. and Mrs. Herrera made donations of their conjugal property to the following: Date of Donation Jan. 25 April 8 May 13 June 13 Sept. 20 Dec. 25 Donee To son, Leonard and his bride, Pam on their...
-
Write a report to Sam By addressing those points below The report should be 2000 words Petty cash is $2000. The cash is used for office supplies, lunches, and other minor expenses. The receipts...
-
Information graphics, also called infographics, are wildly popular, especially in online environments. Why do you think infographics continue to receive so much attention? How could infographics be...
-
A gaseous hydrocarbon (containing C and H atoms) in a container of volume 20.2 L at 350 K and 6.63 atm reacts with an excess of oxygen to form 205.1 g of CO2 and 168.0 g of H2 O. What is the...
-
Chemical tests of four metals A, B, C, and D show the following results. (a) Only B and C react with 0.5 M HCl to give H2 gas.
-
A gas evolved from the fermentation of glucose is found to effuse through a porous barrier in 15.0 min. Under the same conditions of temperature and pressure, it takes an equal volume of N2 12.0 min...
-
Which is an advantage to an employee who participates in a profit-sharing plan? A. Employee does not have to make investment decisions. B. Graded vesting schedule. C. Older employees receive the...
-
Which of the following is a defined benefit plan? A. 403(b). B. 412(i). C. 457(b). D. 457(f).
-
An SEP has which characteristic? A. Loans and hardship withdrawals are available. B. Age-weighting or cross-testing is permitted. C. Social Security integration is permitted. D. Employer matching is...

Study smarter with the SolutionInn App


