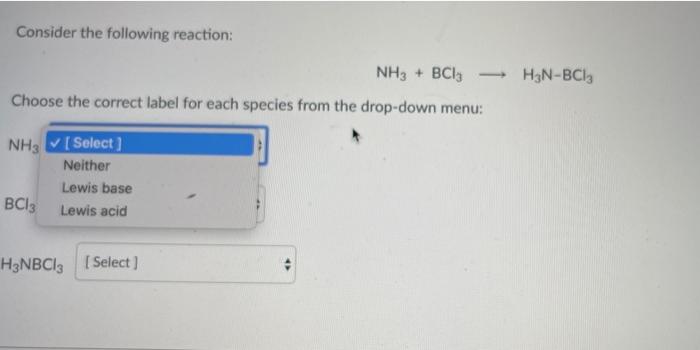
Consider the following reaction: NH3 + BCI3 H3N-BCI3 Choose the correct label for each species from...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following reaction: NH3 + BCI3 H3N-BCI3 Choose the correct label for each species from the drop-down menu: NH3 [Select] Neither Lewis base BCI3 Lewis acid H3NBCI3 (Select] Consider the following reaction: NH3 + BCI3 H3N-BCI3 Choose the correct label for each species from the drop-down menu: NH3 [Select] Neither Lewis base BCI3 Lewis acid H3NBCI3 (Select]
Expert Answer:
Answer rating: 100% (QA)
1 NH3 Lewis base 2 BCl3 Lewis acid 3 H3NBCl3 Neither BCl3 molecule accepts ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction between mercury (II) chloride and oxalate ion: 2 HgCI2(aq) + C2O42- (aq) 2 CIË (aq) + 2 CO2(g) + Hg2CI2(s) The initial rate of this reaction was determined for...
-
Consider the following reaction between oxides of nitrogen: NO2(g) + N2O(g) 3 NO (g) (a) Use data in Appendix C to predict how G for the reaction varies with increasing temperature. (b) Calculate G...
-
Ultimate Consulting is a management consulting firm that offers a wide range of services to companies in many industries. To stay on the cutting edge of developments in the business world and to...
-
Consider the analysis performed in Example 8.6.2. This time, use the usual improper prior to compute the posterior distribution of the parameters.
-
Data for Decorators Mill Ltd. were presented in E3.6. In E3.6. Mar. 2 Issued common shares for $11,000 cash. 4 Purchased used car for $1,000 cash and $9,000 on account, for use in the business. 10...
-
Tom was the manager of three research and development laboratories for a large chemical and materials corporation. He supervised general operations, budgeting, personnel, and proposal development for...
-
Multinational transfer pricing, global tax minimization The Mornay Company manufactures telecommunications equipment at its plant in Toledo, Ohio. The company has marketing divisions throughout the...
-
What link should a user with Sign/Submit access select from the sidebar menu when preparing to submit a revised subapplication?
-
One way to see whether this procedure will be successful is to split the original data set into two subsets: one subset for estimation and one subset for validation. A regression equation is...
-
The following information applies to the questions displayed below Juan Diego began the year with a tax basis in his partnership interest of $50,000 During the year, he was allocated $20,000 of...
-
2. Assume that you are considering investing in two risky assets with the following probability distribution. Stock State of the World Probability 1 0.30 2 FLI CEB Return (%) Return (%) 8 0 5 -1 5 2...
-
Considering the wars, terrorist attacks, and other violations of international law around the world, discuss the function of international law in regulating the relations between sovereign nations in...
-
What is the Present Value for $15,000 in 30 years at discount rate of 12%? XYZ mutual fund has an average 6% annual rate of return. How much money you could make in 20 years if you invest 80,000 at...
-
Why not think that women are better able to reflect critically on masculinity and male experience than men since they do not need to imagine the perspective of the oppressed? I think there is a lot...
-
Sea P(A) = 0,52, P(B) = 0,27 y P(A|B) = 0,47. una. Calcular P(ANB) b. Calcular P(AUB) C. Calcular P(BIA)
-
Solution 9 Pw = 1000kg/m SG = fw = 0.5m 7/5x092 = 4.6. 8lokg/m Toookg/m 3 b = wookg/m v = 54104m/s 2= VP = lookg/mx5x10m/5x0.92 Solution: P+PAir + Parastihe h [1m =farm +/googens h 1300+12.054 +6664...
-
Is the modified 5-question approach to ethical decision making superior to the modified moral standards or modified Past in approach?
-
What is the electron configuration for the transition metal ion(s) in each of the following compounds? a. (NF4)2[Fe(h2O)2CI4] b. [Co(NH3)2(NH2CH2CH2NH2)2]I2 c. Na2[TaF7] d. [Pt(NH3)4I2][Ptl4] Pt...
-
Without looking at data in the text, sketch a qualitative graph of the third ionization energy versus atomic number for the elements Na through Ar, and explain your graph.
-
Although y rays are far more penetrating than a particles, the latter are more likely to cause damage to an organism. Why? Which type of radiation is more effective at promoting the ionization of...
-
Kids Sports Consulting Pty Ltd is a company set up by sports and recreation management students to gain experience in running their own business. It had the following contribution margin income...
-
V. Zarb, the marketing manager for Maltese Treasures Ltd, is preparing a sales budget for the year ended 30 June 2020. In reviewing the actual sales data for the previous year, the sales and...
-
The following expenses budget has been prepared for Abacus Services for the year ending 30 June 2020. Professional salaries, secretarial wages and training are paid in the quarter in which they are...

Study smarter with the SolutionInn App


