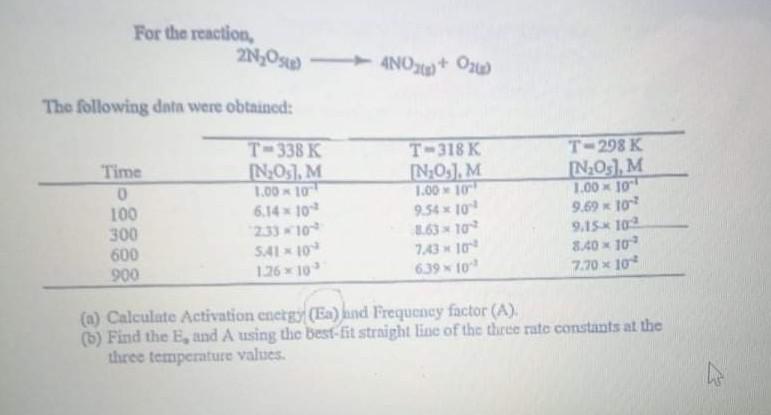
For the reaction, 2NO4NO2+0) The following data were obtained: Time 0 100 300 600 900 T-338...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For the reaction, 2N₂O4NO2+0) The following data were obtained: Time 0 100 300 600 900 T-338 K [N₂0s], M 1.00-10 6.14 × 10 2.33-10 5.41-10 1.26×10³ T-318 K [N₂0₂], M 1.00 10 9.54 × 10¹ 8.63 10² 7.43 × 10¹¹ 6.39 × 10³ T-298 K IN₂0s], M 1.00 10 9.69 × 10² 9.15 102 8.40 10 7.70 × 10² (a) Calculate Activation energy (Ea) and Frequency factor (A). (b) Find the E, and A using the best-fit straight line of the three rate constants at the three temperature values. For the reaction, 2N₂O4NO2+0) The following data were obtained: Time 0 100 300 600 900 T-338 K [N₂0s], M 1.00-10 6.14 × 10 2.33-10 5.41-10 1.26×10³ T-318 K [N₂0₂], M 1.00 10 9.54 × 10¹ 8.63 10² 7.43 × 10¹¹ 6.39 × 10³ T-298 K IN₂0s], M 1.00 10 9.69 × 10² 9.15 102 8.40 10 7.70 × 10² (a) Calculate Activation energy (Ea) and Frequency factor (A). (b) Find the E, and A using the best-fit straight line of the three rate constants at the three temperature values.
Expert Answer:
Answer rating: 100% (QA)
a Calculating Activation Energy Ea and Frequency Factor A The Arrhenius equation is used to calculate the activation energy Ea and frequency factor A ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In problem 8.16, a college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The instructor carried out a hypothesis test and...
-
You have exams in economics and chemistry coming up, and you have 5 hours available for studying. The following table shows the trade-offs you face in allocating the time you will spend studying each...
-
A college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The passing rate for introductory chemistry is 62%. During one...
-
Question 1 of 5 Which statement best illustrates Crusoe's reasons for teaching Friday about his religion? OA. He does not see Friday as a savage but as a soul worthy of saving. B. He thinks Friday...
-
How likely would each of the following be to use competitive bids? (a) A small town that needed a road resurfaced. (b) A scouting organization that needed a printer to print its scouting handbook....
-
Structure of a vortex? (a) Find a solution to the London equation that has cylindrical symmetry and applies outside a line core. In cylindrical polar coordinates, we want a solution of B ? ? 2 ? 2 B...
-
Explain why there must always be some nonzero electric field outside the plates of a parallel-plate capacitor (Figure P26.13a), which means that the idealization shown in Figure P26.13b, with all the...
-
1. Do Sanders, Ono, and Lyons have the right to challenge the annual picnics value in communicating the corporate culture values? 2. How would company management at Exterior Lighting know whether the...
-
Which OSI layer is responsible for connection - oriented communication?
-
Construct a payoff table for the various demand and stocking quantities. Determine the best amount to stock using regret (minimax). A market buys T-bone steaks from a universitys animal science...
-
Research one of the following companies discussed in your textbook: Snapchat, Taco Bell, Karmaloop, 16 Handles, WeChat, Mxit, Saya, and iCow. Alternatively, feel free to research other companies. Why...
-
can you add to this: HP Inc. is a multinational information technology company that provides personal computing and other access devices, imaging and printing products, and related technologies,...
-
4 On 1/1/2022, Illini awarded its key executives 20,000 stock options to purchase 20,000 of its $1 par common shares at $10 per share within next 5 years, but not before 1/1/2024 (the vesting date)....
-
Kelly Company, a small corporation, is preparing its 2016 nancial statements. The CFO is in the process of preparing the statement of cash flows for presentation to the auditors, but has never...
-
A skydiver is jumping off a hot air balloon that is hovering several hundreds of meters above the ground. The balloon is ascending straight up in positive y - direction with a velocity of 3 . 0 0 m /...
-
The mass of a disk of radius R and thickness t is not uniformly distributed; it has density, i.e., mass per unit volume, (r ) = 0/[1 + r 2/R2], where 0 is the density at the center and r is the...
-
In the figure below, determine the point (other than infinity) at which the electric field is zero. (Let q = -2.20 C and 92 = 6.80 C.) m to the left of q1 Need Help? 91 Master It -1.00 m- 92
-
Write the expression in radical notation. Then evaluate the expression when the result is an integer. 23 -1/2
-
Define each of the following. a. Elementary step b. Molecularity c. Reaction mechanism d. Intermediate e. Rate-determining step
-
The radius trend and the ionization energy trend are exact opposites. Does this make sense? Define electron affinity. Electron affinity values are both exothermic (negative) and endothermic...
-
Consider the reaction 3A + B + C D + E where the rate law is defined as An experiment is carried out where [B]0 = [C]0 = 1.00 M and [A]0 = 1.00 Ã 10-4 M. a. If after 3.00 minutes [A] = 3.26...
-
Milj-Spe AS produces two joint products, cooking oil and soap oil, from a single vegetable oil refining process. In July 2022, the joint costs of this process were NOK 24 000 000. Separable...
-
Galinha-Esquina SA grows and processes chickens. Each chicken is disassembled into five main parts. Information pertaining to production in July 2022 is as follows: Joint costs of production in July...
-
Claude Deux-Bussy is the owner and operator of Boissons Barbe-Bleue Sarl, a bulk soft-drink producer. A single production process yields two bulk soft drinks, Pellas (the main product) and Mlisande...

Study smarter with the SolutionInn App


