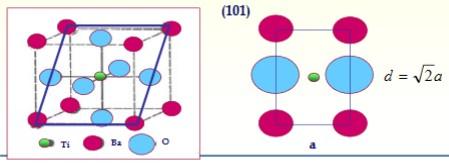
The compound BaTiO 3 has a perovskite structure. Considering the following ratio of radii: (R (O 2-
Fantastic news! We've Found the answer you've been seeking!
Question:
The compound BaTiO3 has a perovskite structure. Considering the following ratio of radii: (R (O2-) = 0.146nm, R (Ti4+ ) = 0.071nm, R (Ba2+) = 0.113nm, Na = 6.023x1023 mol-1, Patm (Ba) = 137.33 g / mol ; Patm (Ti) = 47.88 g / mol; Patm (O) = 15.99 g / mol;
b) Determine the lattice parameters of the unit cell
c) Determine the planar density in the (101) plane (in ions / nm2)
d) Determine the volumetric density (in g / cm3)
Related Book For 

Introduction to Probability
ISBN: 978-0716771098
1st edition
Authors: Mark Daniel Ward, Ellen Gundlach
Posted Date:




