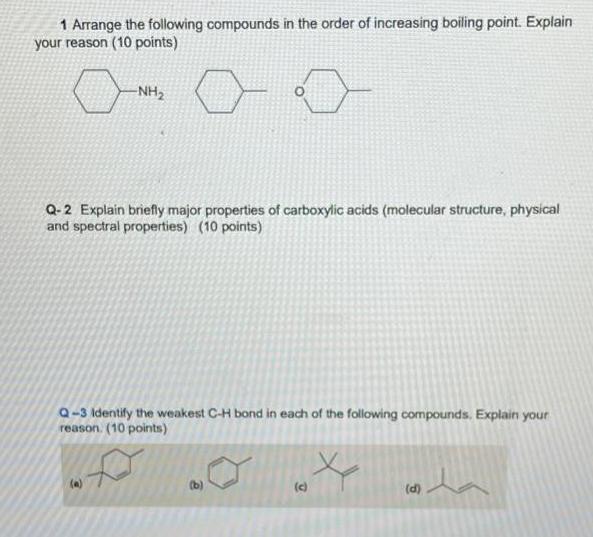
1 Arrange the following compounds in the order of increasing boiling point. Explain your reason (10...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1 Arrange the following compounds in the order of increasing boiling point. Explain your reason (10 points) -NH2 Q- 2 Explain briefly major properties of carboxylic acids (molecular structure, physical and spectral properties) (10 points) a-3 Identify the weakest C-H bond in each of the following compounds. Explain your reason. (10 points) (a) (b) () (d) 1 Arrange the following compounds in the order of increasing boiling point. Explain your reason (10 points) -NH2 Q- 2 Explain briefly major properties of carboxylic acids (molecular structure, physical and spectral properties) (10 points) a-3 Identify the weakest C-H bond in each of the following compounds. Explain your reason. (10 points) (a) (b) () (d)
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
In problem 8.16, a college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The instructor carried out a hypothesis test and...
-
You have exams in economics and chemistry coming up, and you have 5 hours available for studying. The following table shows the trade-offs you face in allocating the time you will spend studying each...
-
A college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The passing rate for introductory chemistry is 62%. During one...
-
Use the following transactions to answer the questions. Use a 6% sales tax rate. Total and rule the sales journal. Jan. 3 Sold merchandise on account to J. J. Johnson, $1,500, plus sales tax. 7 Sold...
-
On December 31, 2016, Vail Company owned the following assets: Vail computes depreciation and amortization expense to the nearest whole year. During 2017, Vail engaged in the following transactions:...
-
Consider the circuit shown below. Write equations for the three currents in terms of R and V . (Indicate the direction with the signs of your answers.) I 1 = I 2 = I 3 = 12 13 R 2R 2R V V
-
Refer to the Arctic Springs Filtration Department Data Set. At Arctic Springs, water is added at the beginning of the filtration process. Conversion costs are incurred evenly throughout the process,...
-
QP Corp. sold 4,000 units of its product at $ 50 per unit in year 2013 and incurred operating expenses of $ 5 per unit in selling the units. It began the year with 700 units in inventory and made...
-
1. How the singly linked lists can be represented? 2. How the doubly linked list can be represented? 3. What are benefits of ADT? 4. When singly linked list can be represented as circular linked...
-
A small island nation is endowed with indestructible coconut trees. These trees live forever and no new trees can be planted. Every year $1 million worth of coconuts fall off the trees and can be...
-
A piece of insulation consists of a layer of material A and a layer of material B, each of thickness L, but with different thermal conductivities. Suppose that the insulation separates a reservoir at...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...
-
completing Emotional Intelligence Skills Assessment in the EISA booklet by Stein, Mann and Papadogiannis ; After reading about the theory behind emotional intelligence and taking the assessment,...
-
Obtain an exponential function in the form f(t) = Aert. f(t) = f(t) is the value after t years of a $5,000 investment earning 8.6% interest compounded continuously.
-
1) specify the discount rate, for the calculation of the Present value of after-tax operating, cash flow Present value of depreciation tax shield Present value of the salvage value of molding...
-
Part 5c: Describe one historical personality involved in the California Mission system, and explain his or her significance in this event. Why was that person significant? Compose your answer in at...
-
Before Gaby bought a camera, she reviewed magazines, solicited opinions from friends and families, conducted online searches, and tested out different cameras in the store. This is an example of a(n)...
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
Predict the product of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) HBr (excess), OH (1) NaH OH Br2 (excess) H20 H3C OH excess QH (1) NaH,Br (2) heat HNO3, H2SO4 NaNH2, NHa (1) NaOH...
-
Consider the interconversion of cis-2-butene and trans-2-butene. (a) What is the value of Ho for the reaction cis-2-butene : trans-2-butene? (b) Assume Ho Go. What minimum value of DG would you...
-
The example of a silyl-Hilbert-Johnson nucleosidation reaction in Section 25.3 is presumed to involve an intermediate ribosyl cation that is stabilized by intramolecular interactions involving the C2...
-
Role of controller, role of chief financial officer. George Jimenez is the controller at Balkin Electronics, a manufacturer of devices for the computer industry. The company may promote him to chief...
-
In the following, assume that all growth and discount rates are stated in real terms. 1. Assume the Eurozone inflation-adjusted average growth in capital stock is 3.0 percent per annum into...
-
Which conclusion presented by Ryan about the top-down approach and the bottom-up approach is most likely correct? A. Conclusion 1. B. Conclusion 2. C. Conclusion 3. Use the following information to...

Study smarter with the SolutionInn App


