Classify each of the following hydrocarbons as an alkane, alkene, or alkyne. Drag the appropriate hydrocarbons...
Fantastic news! We've Found the answer you've been seeking!
Question:
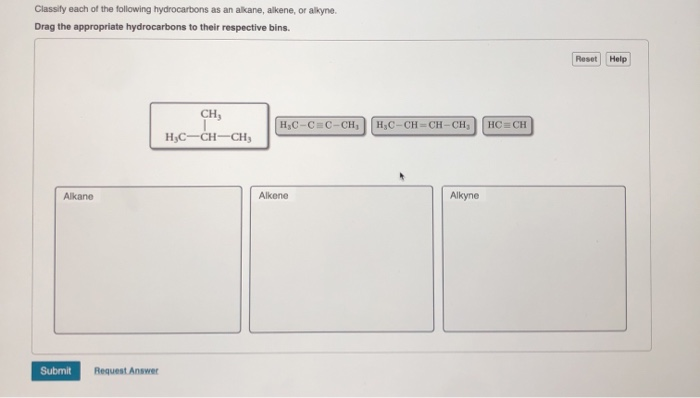
Transcribed Image Text:
Classify each of the following hydrocarbons as an alkane, alkene, or alkyne. Drag the appropriate hydrocarbons to their respective bins. Alkane Submit Request Answer CH, H₂C-CH-CH₂ H₂C-CC-CH₁ H₂C-CH=CH-CH₂ Alkene Alkyne HC CH Reset Help Classify each of the following hydrocarbons as an alkane, alkene, or alkyne. Drag the appropriate hydrocarbons to their respective bins. Alkane Submit Request Answer CH, H₂C-CH-CH₂ H₂C-CC-CH₁ H₂C-CH=CH-CH₂ Alkene Alkyne HC CH Reset Help
Expert Answer:
Answer rating: 100% (QA)
Answer Alkanes are saturated compounds that means they have only single ... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Write methods rotateLeft and rotateRight that rotate the pixels of an image counter-clockwise or clockwise by 90 degrees respectively. You should not assume that the image is square in shape; its...
-
Classify each of the following statements as true or false: (a) Although both molecular solids and covalent-network solids have covalent bonds, the melting points of molecular solids are much lower...
-
Classify each of the following statements as true or false: (a) For molecular solids the melting point generally increases as the strengths of the covalent bonds increase. (b) For molecular solids...
-
Ethelbert.com is a young software company owned by two entrepreneurs. It currently needs to raise $400,000 to support its expansion plans. A venture capitalist is prepared to provide the cash in...
-
Tonys favorite memories of his childhood were the times he spent with his dad at camp. Tony was daydreaming of those days a bit as he and Suzie jogged along a nature trail and came across a wonderful...
-
Beginning inventory, purchases, and sales for WCS12 are as follows: Assuming a perpetual inventory system and using the weighted average method, determine (a) The weighted average unit cost after the...
-
In December 2008, Jason Garcia signed a motor vehicle sales contract with Mac Haik Dodge Chrysler Jeep, a dealer. In the contract, Garcia agreed to purchase a 2009 Dodge Ram 1500. The contract...
-
Managers at Wagner Fabricating Company are reviewing the economic feasibility of manufacturing a part that it currently purchases from a supplier. Forecasted annual demand for the part is 3200 units....
-
Given a public good with supply given by P = 2 + Q, and 4 consumers, each with demand of P = 15.5 - Q, find the optimal aggregate CS (Answer 1) and PS (Answer 2). Blank # 1 Blank # 2 A
-
Gloria and Deloria, CPAs, have recently started their public accounting firm and intend to provide attestation and a variety of consulting services for their clients, which are all nonpublic. Both...
-
Prior to 2019, lessees did not include the right-of-use asset and the lease liability for operating leases on their balance sheets. Both FASB and IASB wrote new standards to require that lessees...
-
You have $52,260.84 in a brokerage account, and you plan to deposit an additional $3,000 at the end of every future year until your account totals $290,000. You expect to earn 14% annually on the...
-
According to the data collected by the Center for Medicare and Medicaid Services, health spending in the United States as a percentage of GDP has risen from 5% in 1960 to 18% in 2019. Which of the...
-
What are the most important concepts, theories, tools... you have learned so far in this class? 2 . What information don't you think you understand well enough yet? 3 . Are there any technology...
-
In relation to Australian Law can you explain the concept of lifting the corporate veil with reference to examples and relevant law?
-
Consider the wheat market. In this market both the demand and the supply curves are linear. When the price is Rs.15, the quantity sold is 15kg and there is a surplus of an additional 15kg wheat....
-
Hong Publishing has purchased Lang Publishing. After reviewing titles from both companies, a decision must be made to determine what titles must be dropped. The following information is available to...
-
You are a U.S. investor who purchased British securities for 2,000 one year ago when the British pound cost U.S. $1.50. What is your total return (based on U.S. dollars) if the value of the...
-
R)-(+)-Glyceraldehyde can also be transformed into (-)-malic acid. This synthesis begins with the conversion of (R)-(+)-glyceraldehyde into (-)-tartaric acid, as shown in Problem 17.41, parts (e) and...
-
There are four amides with the formula C3H7NO. (a) Write their structures. (b) One of these amides has a melting and a boiling point that are substantially lower than those of the other three. Which...
-
When the compound called isoborneol is heated with 9 M sulfuric acid, the product of the reaction is the compound called camphene and not bornylene, as one might expect. Using models to assist you,...
-
Over what ranges of taxable income in 2017 will the total income tax liability for two persons with equal incomes who file as single individuals equal their income tax liability if they file jointly...
-
If a taxpayer has $40,000 of employee salary in 2017, how much will be withheld for the Social Security and Medicare taxes?
-
Sally and Jim are married and have taxable income in 2017 of $160,000. If they could file their income tax as single individuals, each of them would have taxable income of $80,000. Do they have a...

Study smarter with the SolutionInn App


