Classify each of the molecules according to its functional group. alcohol alcohol ketone carboxylic acid aldehyde...
Fantastic news! We've Found the answer you've been seeking!
Question:
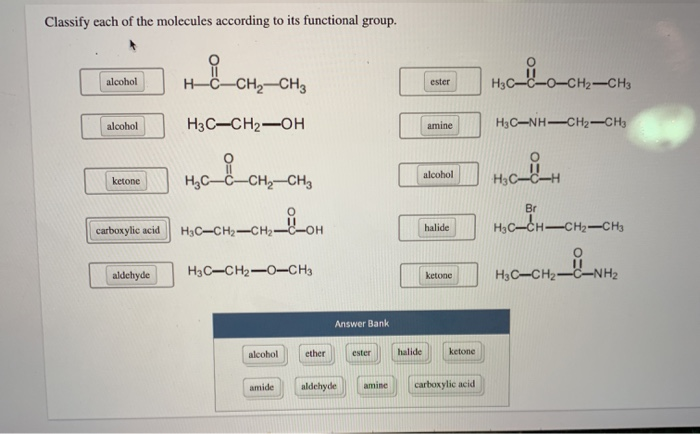
Transcribed Image Text:
Classify each of the molecules according to its functional group. alcohol alcohol ketone carboxylic acid aldehyde HC CH, CH H3C-CH₂-OH навононо H₂C-CH₂-CH₂-OH H3C-CH₂-O-CH3 alcohol amide ether Answer Bank ester aldehyde amine halide ester amine alcohol halide ketone ketone carboxylic acid H3C-C-O-CH2-CH3 H3C—NH–CH2–CH3 H₂C_H -H Br HỌC—CH—CH2–CH3 NH H3C-CH₂- Classify each of the molecules according to its functional group. alcohol alcohol ketone carboxylic acid aldehyde HC CH, CH H3C-CH₂-OH навононо H₂C-CH₂-CH₂-OH H3C-CH₂-O-CH3 alcohol amide ether Answer Bank ester aldehyde amine halide ester amine alcohol halide ketone ketone carboxylic acid H3C-C-O-CH2-CH3 H3C—NH–CH2–CH3 H₂C_H -H Br HỌC—CH—CH2–CH3 NH H3C-CH₂-
Expert Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Classify each of the following heterocyclic molecules as aromatic or not, according to Huckel's rule: HN HB BH 0. NH
-
Classify each of the following species as a Lewis acid or a Lewis base: (a) CO2, (b) H2O, (c) I-, (d) SO2, (e) NH3, (f) OH-, (g) H+, (h) BCl3.
-
Classify each of the following heterocyclic molecules as aromatic or not, according to Hackles rule:
-
A city starts a solid waste landfill in 2012. When the landfill was opened the city estimated that it would fill to capacity within 5 years and that the cost to cover the facility would be $1.5...
-
During 2014, Alpha, Inc., had sales of $ 6.86 billion, operating profit of $ 2.20 billion, and net income of $ 3.40 billion. EPS was $ 6.75. On May 11, 2015, one share of Alphas common stock was...
-
RPR, Inc., anticipates that 120,000 units of product K will be sold during May. Each unit of product K requires four units of raw material A. Actual inventories as of May 1 and budgeted inventories...
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
An investment advisor at Shore Financial Services wants to develop a model that can be used to allocate investment funds among four alternatives: stocks, bonds, mutual funds, and cash. For the coming...
-
Consider the following logarithmic equation. log3(x) log.x 8- log(x) log(x) = 1. (i) Find the value(s) of a satisfying in the equation. (ii) Determine for what values of the logarithmic expression on...
-
1. 3. 5. 7. 9. 11. passive : change:: active: O movement O increase O flight stagnation convent: cloister :: eyrie: O hide nest O open show rotate: energy :: stop: O brake Ostart Orun push fumble:...
-
A square shaped wire loop with a side length of a = 36.1 cm carries an electric current of I = 16.9 A as shown in the figure. a)Determine the size of the magnetic field at point P, the center of the...
-
Consumers make countless decisions every day. As consumers, we solve problems and fulfill needs such as making lunch plans or getting fuel for the car. Advertising impacts consumer decisions. Respond...
-
Eli Orchid has designed a new pharmaceutical product, Orchid Relief, which improves the night sleep. Before initiating mass production of the product, Eli Orchid has been market-testing Orchid Relief...
-
it was around the 1660s when the fundamental theories that would become the study of calculus were underway. In Germany, a man named Gottfried Wilhelm Leibniz studied calculus, while in England,...
-
The expected return on a stock X is said to be E(X)-4% and the variance on the return V(X) = 0.25 Another stock Y has expetced value and variance E(Y) =6% and V(Y) = 0.49 The correlation between...
-
In one or two sentences, describe how a credit card works. What is the advantage of paying your credit card balance in full each month? What is an outstanding balance? Why is it more difficult to get...
-
You are purchasing a $4,250,000.00 Money Market CD that matures in 363 days and is quoted as a 7.12% interest at maturity. What is the straigh yield if you hold this CD to maturity? 6) You are...
-
Refrigerant-134a enters an adiabatic compressor as saturated vapor at 120 kPa at a rate of 0.3 m3/min and exits at 1-MPa pressure. If the isentropic efficiency of the compressor is 80 percent,...
-
Write the expected products of reaction of 1-ethylcyclopentene with each of the reagents in Problem 46. (a) H 2 , PtO 2 , CH 3 CH 2 OH (b) D 2 , Pd C, CH 3 CH 2 OH (c) BH 3 , THF then NaOH + H 2 O 2...
-
The reaction of CH4 with Cl2 to yield CH3C1 and HCI is well known. On the basis of the values in the short table below, the enthalpy H° (kcal mol-1) of this reaction is (a) +135 (b) -135 (c) +25...
-
Two of the following four compounds are more acidic than CH 3 OH (i.e., two of these have K a greater than methanol). Which ones? (a) A and B; (b) B and C; (c) C and D; (d) D and A; (e) D and B....
-
Aggarwal suggests that young people just starting at work might rely too much on what he calls Level 1 research: jumping on the Internet to find information. Would you consider that a strength of new...
-
Kaplan also notes how time- and labor-intensive it is to prepare proposals and sustain good relationships with funders and others. Which of Kaplans remarks underscore the importance of good business...
-
Which specific paperless tools are discussed by Shelby Walsh? Which are inferior to paper-based media, and which are superior? Is an entirely paperless office possible, or does paper still have...

Study smarter with the SolutionInn App



