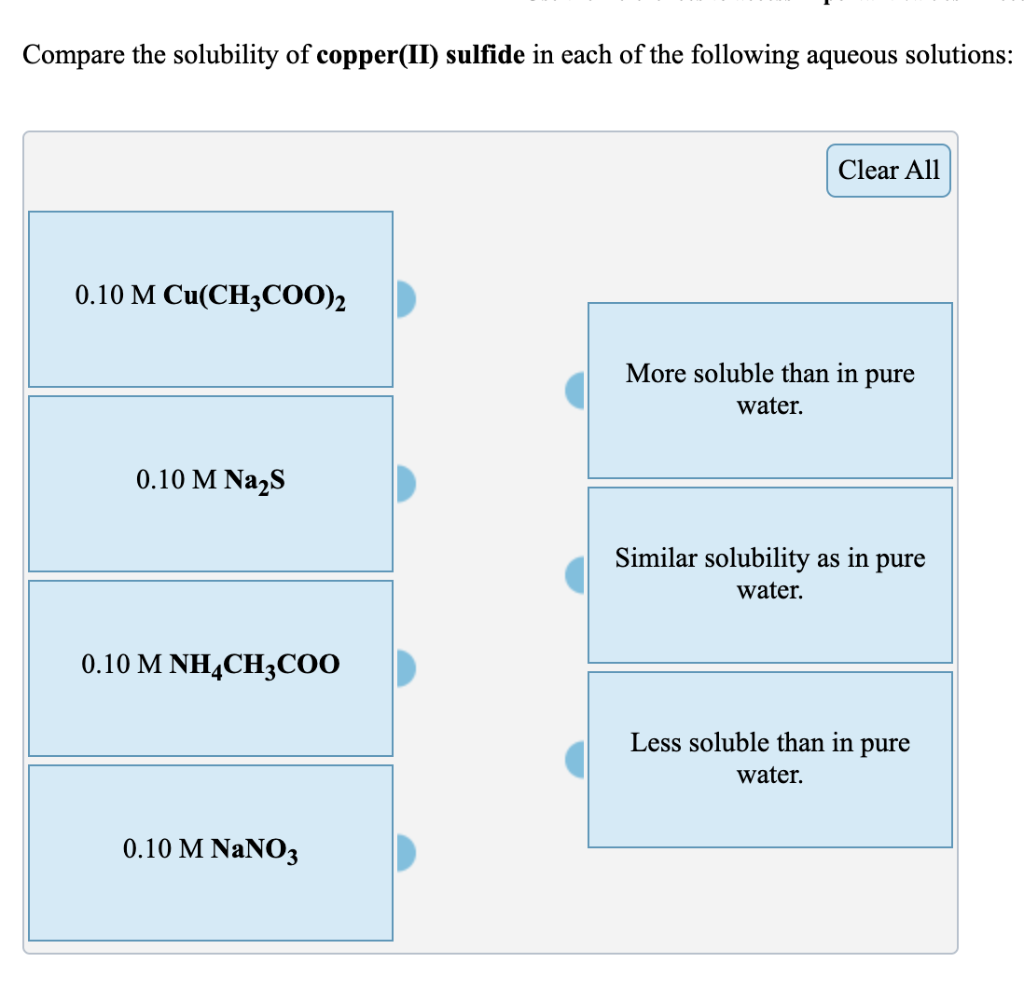
Compare the solubility of copper(II) sulfide in each of the following aqueous solutions: 0.10 M Cu(CH3COO)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
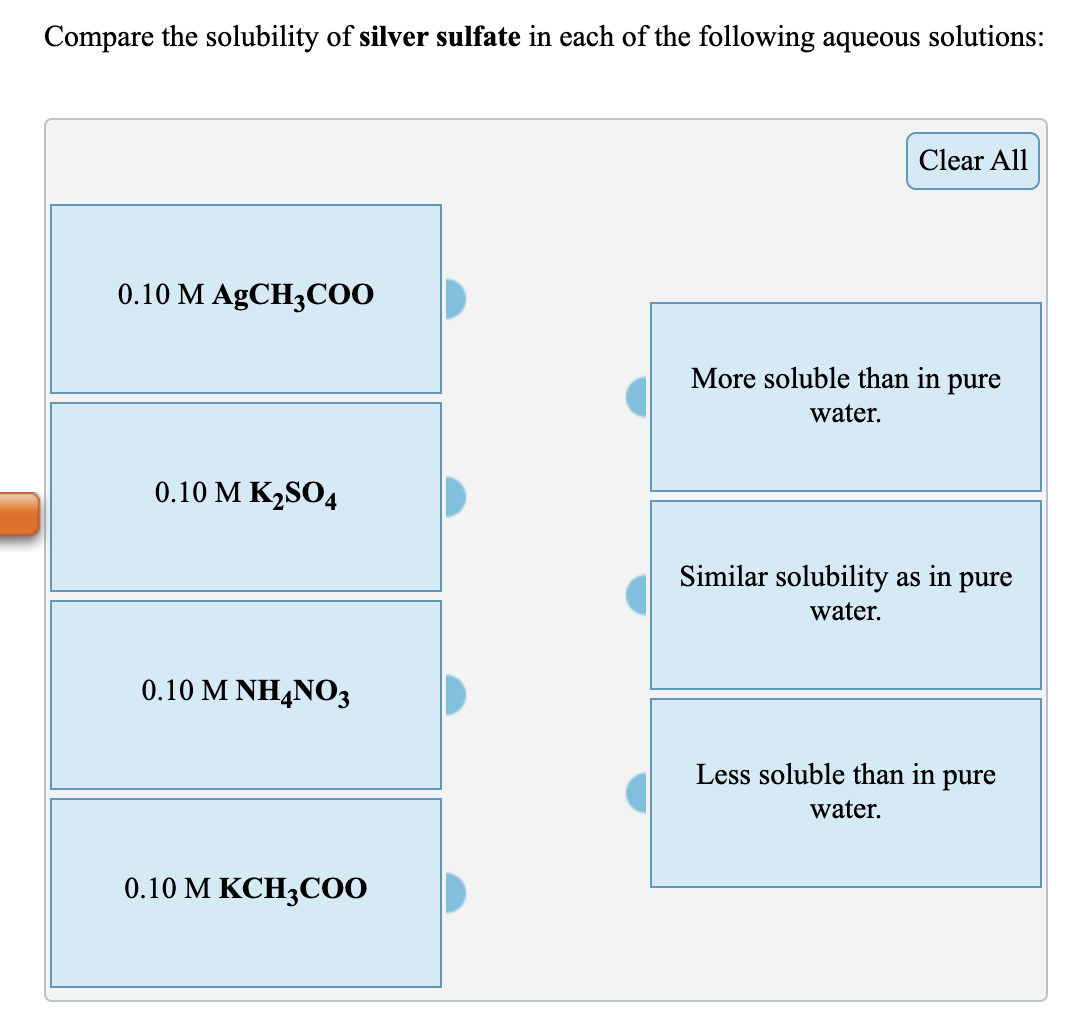
Compare the solubility of copper(II) sulfide in each of the following aqueous solutions: 0.10 M Cu(CH3COO)₂ 0.10 M Na₂S 0.10 M NH4CH3COO 0.10 M NaNO3 Clear All More soluble than in pure water. Similar solubility as in pure water. Less soluble than in pure water. Compare the solubility of silver sulfate in each of the following aqueous solutions: 0.10 M AgCH3COO 0.10 M K₂SO4 0.10 M NH4NO3 0.10 M KCH3COO Clear All More soluble than in pure water. Similar solubility as in pure water. Less soluble than in pure water. Compare the solubility of copper(II) sulfide in each of the following aqueous solutions: 0.10 M Cu(CH3COO)₂ 0.10 M Na₂S 0.10 M NH4CH3COO 0.10 M NaNO3 Clear All More soluble than in pure water. Similar solubility as in pure water. Less soluble than in pure water. Compare the solubility of silver sulfate in each of the following aqueous solutions: 0.10 M AgCH3COO 0.10 M K₂SO4 0.10 M NH4NO3 0.10 M KCH3COO Clear All More soluble than in pure water. Similar solubility as in pure water. Less soluble than in pure water.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Compare the solubility of nickel (II) carbonate in each of the following aqueous solutions: 0.10 M Ni(CH3COO) 0.10 M KCO3 0.10 M KNO3 0.10 M NHCH3COO Clear All More soluble than in pure water....
-
Classify each of the following aqueous solutions as a nonelectrolyte, weak electrolyte, or strong electrolyte: (a) LiClO4 (b) HClO (c) CH3CH2CH2OH (propanol) (d) HClO3 (e) CuSO4 (f) C12H22O11...
-
The room-temperature tensile strengths of pure copper and pure silver are 209 and 125 MPa, respectively. (a) Make a schematic graph of the room-temperature tensile strength versus composition for all...
-
Should U.S. national forests become privatized (sold to private owners)? Why or why not?
-
What is the relationship of audit procedures to assertions that are embodied in financial statement representations?
-
A resistor consists of three identical metal strips connected as shown in Fig. Q26.8. If one of the strips is cut out, does the ammeter reading increase, decrease, or stay the same? Why? Figure Q26.8...
-
How do stakeholder groups relevant to environmental management accounting and environmentally induced financial accounting differ?
-
Under the decimal system of computing time worked at Carman's Company, production workers who are tardy are "docked" according to the schedule shown below. Minutes Late in Ringing inFractional Hour...
-
OIM airline provides 120 seats on its flight. Customers looking for discounts can purchase tickets at $260 per seat. The tickets purchased closer to the flight date are charged $360 per seat,...
-
In addition to providing polarity data, like those you interpreted in Part B, most terrestrial lava flows contain minerals that can be precisely dated using radiometric age-dating techniques. The...
-
Pavin acquires all of Stabler's outstanding shares on January 1, 2018, for $570,000 in cash. Of this amount, $41,000 was attributed to equipment with a 5-year remaining life and $51,000 was assigned...
-
Jargon Jen has five million shares outstanding, generates free cash flows of $12 million each year and has a cost of capital of 6%. It also has $3 million of cash on hand. Jargon Jen wants to decide...
-
Metabolizing a candy bar releases about 1 0 ^ 6 joules of energy. Assume the candy bar's mass is 0 . 3 6 kg and use the formula kinetic energy = 1 / 2 mv ^ 2 , where m is the object's mass and v is...
-
If the children are bouncing down the road - since the buses shocks are so done - what is the frequency of those bounces if the bus + driver + kids mass is 1 2 , 0 8 2 kg and the bus spring constant...
-
You are thinking about buying a printing press today that will print t-shirts for many years including the celebration of a DePaul Women's Soccer National Championship. The machine will cost $5mm to...
-
1 4 . A mixture containing 1 2 . 8 uM analyte ( X ) and 4 4 4 of internal standard ( IS ) gave a ning an chromatographic peak area of 3 0 6 for X and 5 1 1 for IS . A second solution contai unknown...
-
A root sequence is a series of positive numbers for obeying the following rule: where fn-1 is the number before fn. fn=1+ -1 (1) Write two Python scripts, one with for loop and the other with while...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Ammonium chloride, NH4Cl, is a white solid. When heated to 325C, it gives a vapor that is a mixture of ammonia and hydrogen chloride. NH4Cl(s) NH3(g) + HCl(g) Suppose someone contends that the vapor...
-
Explain how ionic radii are obtained from known distances between nuclei in crystals.
-
The lead atom has the groundstate configuration [Xe]4f145d106s26p2. Find the period and group for this element. From its position in the periodic table, would you classify lead as a main-group...
-
What are the advantages of resource leveling?
-
Which of the following statements is false? A. The highest need in Maslows pyramid is called selfactualization. B. Most people today prefer managers who follow Theory X versus Theory Y. C. Herzberg...
-
Some project managers like to assess team performance by using a technique known as MBWA, which stands for _______________. A. management by wondering aloud B. management by wandering around C....

Study smarter with the SolutionInn App


