Compound 10a is an ester with a molecular formula of CsHO). When 10a is allowed to...
Fantastic news! We've Found the answer you've been seeking!
Question:

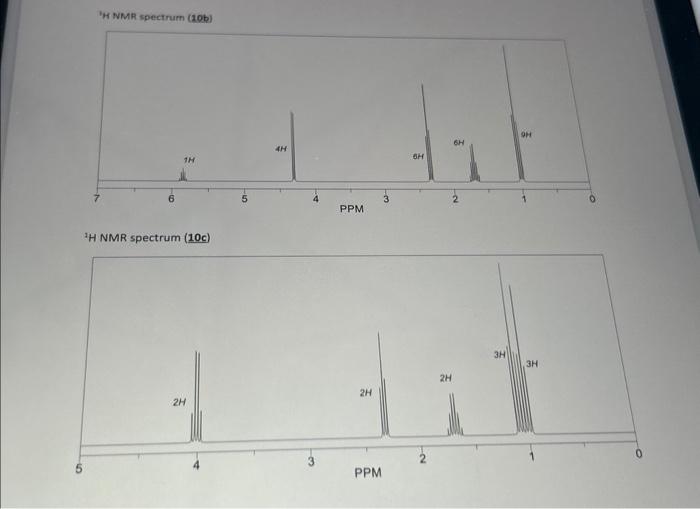
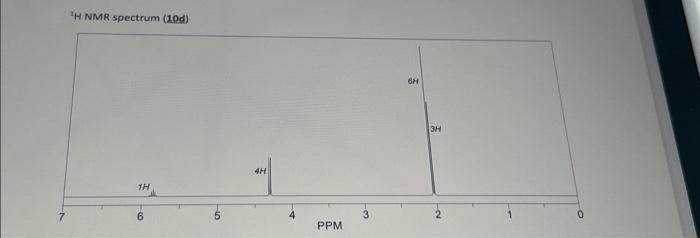
Transcribed Image Text:
Compound 10a is an ester with a molecular formula of CsH₂O). When 10a is allowed to react with glycerin in the presence of an acid catalyst a triester (10b) is generated. This triester reacts remarkably fast when treated with sodium ethoxide in ethyl acetate as the solvent giving two products: 10c (an ester) and 10d (a triester). Using only the provided H NMR spectra (below), give the structures for 10a. 10b. 10c, and 10d. Glycerin + 3 10a cat. H (CsH100₂) (-3 x MeOH)) ¹H NMR spectrum (10a) 3H 10b (C18H2006) 2H excess NaOEt (cat.). EtOA PPM 2H 3 10c (C₂H120₂) 19d (C₂H₂O₂) H NMR spectrum (10b) 1H ¹H NMR spectrum (10c) 2H 4H PPM 2H PPM SH 2H 3H 9H 3H ¹H NMR spectrum (10d) 1H -10 4H PPM -3 6H 3H o Compound 10a is an ester with a molecular formula of CsH₂O). When 10a is allowed to react with glycerin in the presence of an acid catalyst a triester (10b) is generated. This triester reacts remarkably fast when treated with sodium ethoxide in ethyl acetate as the solvent giving two products: 10c (an ester) and 10d (a triester). Using only the provided H NMR spectra (below), give the structures for 10a. 10b. 10c, and 10d. Glycerin + 3 10a cat. H (CsH100₂) (-3 x MeOH)) ¹H NMR spectrum (10a) 3H 10b (C18H2006) 2H excess NaOEt (cat.). EtOA PPM 2H 3 10c (C₂H120₂) 19d (C₂H₂O₂) H NMR spectrum (10b) 1H ¹H NMR spectrum (10c) 2H 4H PPM 2H PPM SH 2H 3H 9H 3H ¹H NMR spectrum (10d) 1H -10 4H PPM -3 6H 3H o
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these finance questions
-
Compound A has a molecular formula of C5H12O and is oxidized by an acidic solution of sodium dichromate to give compound B, whose molecular formula H5SO4 is When compound A is heated with C and D are...
-
A compound has a molecular formula of C8H14O4, and its infrared spectrum contains an intense peak at 1730 cm -1. The 1H NMR spectrum of the compound is shown in Figure 20.11. What is its structure?...
-
A compound has a molecular formula of C8H14O4, and its infrared spectrum contains an intense peak at 1730 cm-1. The 1H NMR spectrum of the compound is shown in Figure 20.11. What is its structure?
-
Each student will interview a manager or an employee (who might be a family member, a friend, or an acquaintance) to determine the extent to which the issues raised in the case are represented in his...
-
Show that where is the anlge between u and F. KK" = 1 (u2/c2) cos2 1-u/c F
-
Consider the longitudinally vibrating uniform beam shown in Figure 7.49. The ends of the beam are supported by linear springs with stiffnesses \(k_{1}\) and \(k_{2}\). Derive the frequency equation,...
-
Adjusting the accounts is the process of a. recording transactions as they occur during the period. b. updating the accounts at the end of the period. C. zeroing out account balances to prepare for...
-
Western Sound Studios records and masters audio tapes of popular artists in live concerts. The performers use the tapes to prepare live albums, CDs, and MP3s. The following account balances were...
-
During the early Covid-19 pandemic in 2020, there was no masks available for sale in grocery stores in US. This was a big problem because the use of masks could have minimized the individual risk of...
-
Refer to the following financial statements for Kodak: Liabilities and shareholders equity Current liabilities Required: Prepare forecasts of its income statement, balance sheet, and statement of...
-
Which of the following is NOT Distance vector Protocol O IGRP O ISIS O EIGRP O RIP v2
-
Judy, an internal auditor at Fermented Fruits, has found evidence of accounting fraud at the company. She is worried about the repercussions of reporting her firm to the SEC. You remind her that...
-
Loans to executives were prohibited by the Sarbanes-Oxley Act of 2002 because: (a) The board of directors granted huge loans to company officers, and then conveniently authorized forgiveness of the...
-
At 300 K and 1 bar, the equilibrium constant for the following reaction: is reported to be 10. What is the equilibrium constant at 300 K and 10 bar? You may assume ideal gas behavior. A(g) + B(g) C(g)
-
A classmate believes that the image you see in a mirror is on the mirror surface rather than behind the mirror. What is something you can do to convince your classmate he is incorrect?
-
White light passing through a prism is dispersed into its constituent colors. Describe how this rainbow of colors can be changed back to white light.
-
Create two objects: a rectangle and a square. Also, create a rectangle pointer named rptr and initialize it to nullptr. Then: point the rptr pointer to the rectangle object and use it to call the...
-
Find the equations of the ellipses satisfying the given conditions. The center of each is at the origin. Passes through (2, 2) and (1, 4)
-
Cubitene is a diterpene present in the defense secretion of a species of African termite. What unusual feature characterizes the joining of isoprene units in cubitene?
-
Write an equation showing the preparation of the following amides from the indicated carboxylic acid derivative: (a) (b) 0 CH3CNHCH3 from an acid anhydride HCN(CH3) from a methyl ester
-
Increased "single-bond character" in a carbonyl group is associated with a decreased carbon- oxygen stretching frequency. Among the three compounds benzaldehyde, 2,4,6 trimethoxybenzaldehyde, and...
-
The following are cutoff values for the upper 5% of a \(t\)-distribution with either degrees of freedom 10, 50 , or 100: \(2.23,1.98\), and 2.01. Identify which value belongs to which distribution...
-
Researchers interested in lead exposure due to car exhaust sampled the blood of 52 police officers subjected to constant inhalation of automobile exhaust fumes while working traffic enforcement in a...
-
Suppose that you are interested in determining whether exposure to the organochloride DDT, which has been used extensively as an insecticide for many years, is associated with breast cancer in women....

Study smarter with the SolutionInn App


