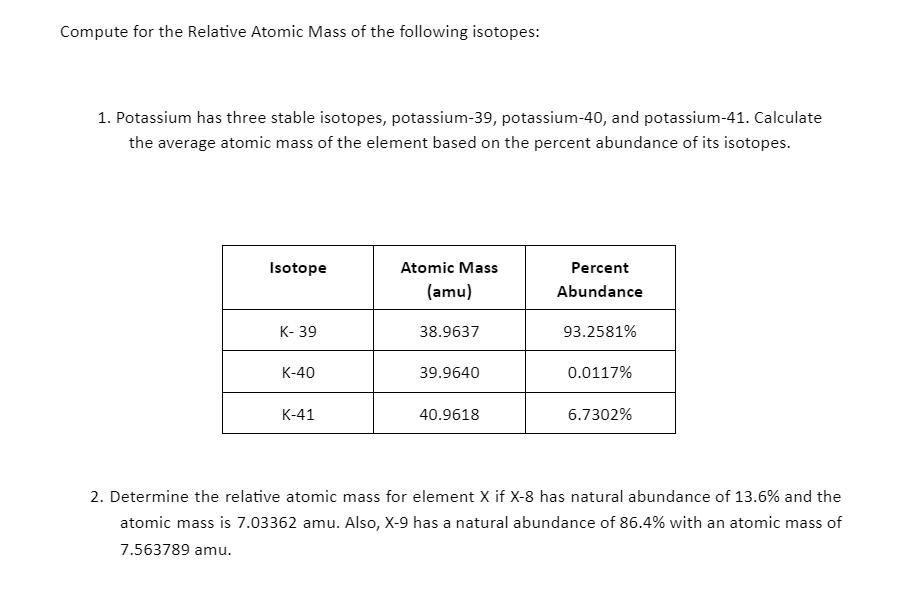
Compute for the Relative Atomic Mass of the following isotopes: 1. Potassium has three stable isotopes,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Compute for the Relative Atomic Mass of the following isotopes: 1. Potassium has three stable isotopes, potassium-39, potassium-40, and potassium-41. Calculate the average atomic mass of the element based on the percent abundance of its isotopes. Isotope K-39 K-40 K-41 Atomic Mass (amu) 38.9637 39.9640 40.9618 Percent Abundance 93.2581% 0.0117% 6.7302% 2. Determine the relative atomic mass for element X if X-8 has natural abundance of 13.6% and the atomic mass is 7.03362 amu. Also, X-9 has a natural abundance of 86.4% with an atomic mass of 7.563789 amu. Compute for the Relative Atomic Mass of the following isotopes: 1. Potassium has three stable isotopes, potassium-39, potassium-40, and potassium-41. Calculate the average atomic mass of the element based on the percent abundance of its isotopes. Isotope K-39 K-40 K-41 Atomic Mass (amu) 38.9637 39.9640 40.9618 Percent Abundance 93.2581% 0.0117% 6.7302% 2. Determine the relative atomic mass for element X if X-8 has natural abundance of 13.6% and the atomic mass is 7.03362 amu. Also, X-9 has a natural abundance of 86.4% with an atomic mass of 7.563789 amu.
Expert Answer:
Answer rating: 100% (QA)
1 The average atomic mass of an element is calculated by taking the weighted average of the masses o... View the full answer

Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these chemistry questions
-
An element consists of 1.40% of an isotope with mass 203.973 amu, 24.10% of an isotope with mass 205.9745 amu, 22.10% of an isotope with mass 206.9759 amu, and 52.40% of an isotope with mass 207.9766...
-
Calculate the atomic mass of an element with two naturally occurring isotopes, from the following data: What is the identity of element X? Fractional Isotope Isotopic Mass (amu) Abundance X-63 X-65...
-
Determine the atomic mass of ruthenium from the given abundance and mass data Ruthenium-96 Ruthenium-98 Ruthenium-99 Ruthenium-100 Ruthenium-101 Ruthenium-102 Ruthenium-104 5.54% 1.87% 12.76% 12.60%...
-
Which of the following statements are true about REST? Pick ONE OR MORE options Logical URLs should be used instead of physical URLS Adal URLs must always be used in REST response A paging technique...
-
Evaluate loss of control as a disadvantage of franchising from the franchisors perspective.
-
What is the entry for Zakah payment made by the IFI on behalf of the shareholders? a. Dr. Zakah expense Cr. Cash/Bank b. Dr. Dividends payable Cr. Cash/Bank c. Dr. Cash/Bank Cr. Dividends payable d....
-
Aaron Reed, a photographer, was in a dispute with Ezelle Investment Properties, Inc., over Ezelle allegedly using one of Reeds photographs without permission. Reed sent Ezelle a cease-and-desist...
-
Machinery purchased for $52,000 by Carver Co. in 2008 was originally estimated to have a life of 8 years with a salvage value of $4,000 at the end of that time. Depreciation has been entered for 5...
-
Jobs R Us, Inc. Is a recruiting firm that specializes in post- college placement in the finance industry. Its clients are currently concentrated in the North-Eastern United States lt is contemplating...
-
Occupy Mall Street Occupy Mall Street (OMS or the Company) is a leading real estate management firm that owns and manages over 100 shopping malls across the United States. The Company went public in...
-
The BPC Company must decide among the following three mutually exclusive investment projects. Each project has a positive NPV and each project's MIRR > WACC. Which invest project should BPC...
-
A rock thrown with speed 1 2 . 0 m / s and launch angle 3 0 . 0 ( above the horizontal ) travels a horizontal distance of d = 1 7 . 5 m before hitting the ground. From what height was the rock...
-
"What is the role of strategic intent and visioning in setting aspirational goals, inspiring organizational commitment, and mobilizing resources towards achieving transformative outcomes?...
-
Mandated nurse-to-patient ratios affect potentially all hospitals and spark much debate in terms of their merits and feasibility of enforcement. The ratio itself is not precise and is influenced by...
-
(30 pts.) Given the four data points (-1,1), (0,1), (1,2),(2,0), determine the interpolating cubic polynomial using the monomial basis; using the Lagrange basis; using the Newton basis. Show that...
-
5. The sun has a radius of 7.0x105 km and spins (rotates) around its central axis with a period of 25 days. What would be the new period if the sun was to shrink (decrease in size), under the action...
-
4 0 DA 4 V3 6 6 Let V = -6 0 , V 12 Is p in Nul A? 1 - 12 , and p = 7. Determine if p is in Nul A, where A = V V V3 BA= [V V V]. -6
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Benzene reacts with bromine. a. Write a balanced chemical equation for this reaction. b. Name the catalyst used. c. What visual observations could be made during the reaction? d. Benzene will also...
-
a. Write equilibrium expressions for the solubility products of the following: i. Fe(OH) 2 ii. Fe 2 S 3 iii. Al(OH) 3 b. State the units of solubility product for each of the compounds in part a.
-
a. Explain why a cation with a smaller ionic radius has a higher charge density. b. Which one of the following ions will be the best polariser of the large nitrate ion? Explain your answer. Cs + Li +...
-
In recent years, Keflavik Paper Company has been having problems with its project management process. Several commercial projects, for example, have come in late and well over budget, and product...
-
Your firm has the opportunity to invest $75,000 in a new project opportunity but due to cash flow concerns, your boss wants to know when you can pay back the original investment. Using the discounted...
-
Pracht, a popular cosmetics company in Germany, has been performing favorably and growing at a constant rate, with profits exceeding $900,000 in the last financial year. The company now wants to...

Study smarter with the SolutionInn App


