Concentration of HSO4 and NaSO4 in a solution is 1 M and 1.810^-2 M, respectively. Molar...
Fantastic news! We've Found the answer you've been seeking!
Question:
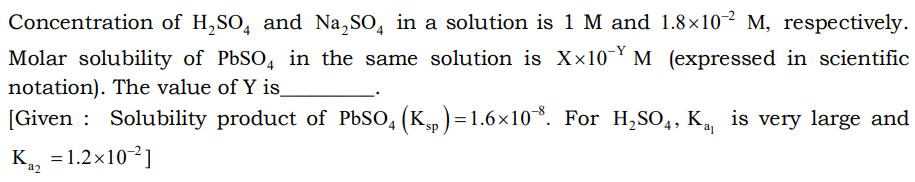
Transcribed Image Text:
Concentration of H₂SO4 and Na₂SO4 in a solution is 1 M and 1.8×10^-2 M, respectively. Molar solubility of PbSO4 in the same solution is X×10 M (expressed in scientific notation). The value of Y is [Given: Solubility product of PbSO4 (Ksp)=1.6×108. For H₂SO4, K₁, is very large and K₁2 = 1.2×10-2] a2 Concentration of H₂SO4 and Na₂SO4 in a solution is 1 M and 1.8×10^-2 M, respectively. Molar solubility of PbSO4 in the same solution is X×10 M (expressed in scientific notation). The value of Y is [Given: Solubility product of PbSO4 (Ksp)=1.6×108. For H₂SO4, K₁, is very large and K₁2 = 1.2×10-2] a2
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In Problems 47-50, convert each number to scientific notation and simplify. Express the answer in both scientific notation and in standard decimal form. 48. 50. (60,000) (0.000003)...
-
The molar heat capacities for carbon dioxide at 298.0 K are Cv = 28.95 J K-1 mol-1 Cp = 37.27 J K-1mol-1 The molar entropy of carbon dioxide gas at 298.0 K and 1.000 atm is 213.64 JK-1mol-1. a....
-
A regression of x = tannin concentration (mg/L) and y = perceived astringency score was considered in Examples 5.2 and 5.6. The perceived astringency was computed from expert tasters rating a wine on...
-
Figure (a) shows a mobile hanging from a ceiling; it consists of two metal pieces (m 1 = 3.5) kg and m 2 = 4.5kg) that are strung together by cords of negligible mass. What is the tension in?? (a)...
-
The following represents a recent income statement for Boeing Company. It includes only five separate numbers (two of which are in billions of dollars), two subtotals, and the net earnings...
-
Whiley Company issued a $100,000, fiveyear, 10 percent note to Security Company on January 2, 2016. Interest was to be paid annually each December 31. The stated rate of interest reflected the market...
-
Find the z-score that corresponds to each value. The scores for the reading portion of the ACT test are normally distributed. In a recent year, the mean test score was 21.2 and the standard deviation...
-
Palisade Creek Co. is a merchandising business that uses the perpetual inventory system. The account balances for Palisade Creek Co. as of May 1, 2019 (unless otherwise indicated), are as follows:...
-
Martin Stephens has asked you to develop a document flowchart for the cash receipts system at M and S Trading Pty Ltd. Martin's narrative of the system is as follows: Customer payments include cash...
-
On February 1, 2024. Arrow Construction Company entered into a three-year construction contract to build a bridge for a price of $8,300,000. During 2024, costs of $2,100,000 were incurred with...
-
1) Mary is a DJ and drives a late model Ford Transit van. But she'd like to drive something that makes more of a statement than a white van when she isn't working. But she doesn't want to sell the...
-
A fuel consisting of 80% C12H36 and 20% C14H30 is burned with 30% excess air. The flue gas is at atmospheric pressure. Find the minimum exhaust temperature to avoid condensation.
-
if your company is operating in Ukraine right now, what measures can you take to deal with the relevant risks? Assume that your business is a business service, such as banking, insurance, or...
-
Discuss revenue generating sources at a typical airport. What are the typical sources of revenue? How might the airport manager generate income sources outside these areas? Provide examples.
-
Summarize the difference between men and women and relationships among gender, culture and communication.?
-
ich of the following statements is a form of inductive reasoning known as causal reasoning? Question 12 options: a) According to a recent survey, most people prefer Greek yogurt over traditional...
-
on 8 For the following set of lengths 130, 170, 160, 160, 150, 190 Third quartile is: et red d out of Select one: O a. 160 a question O b. 145 O c. 175 O d. 180
-
The temperature of 2.5 L of a gas initially at STP is raised to 250C at constant volume. Calculate the final pressure of the gas in atm.
-
Photosynthesis produces glucose, C6H12O6 , and oxygen from carbon dioxide and water: (a) How would you determine experimentally the (H°rxn value for this reaction? (b) Solar radiation produces...
-
Calculate DG and KP for the following processes at 25C: (a) H2(g) + Br2(l) 2HBr(g) (b) 1/2 H2(g) + 12 Br2(l) HBr(g) Account for the differences in DG and KP obtained for (a) and (b).
-
Which of the following is not a reason for having decentralized organizations? a. Better information at the local level leads to su perior decisions. b. Goal congruence is enhanced. c. Quicker...
-
A cost center is responsible for which of the following? a. Investing in long-term assets. b. Controlling costs. c. Generating revenues. d. All of the above.
-
An investment center is responsible for: a. Investing in long term assets. b. Controlling costs. c. Generating revenues. d. All of the above.

Study smarter with the SolutionInn App



