2.5, Consider a mixture of 25% ammonia, the rest nitrogen and hydrogen in a 1:3 ratio....
Fantastic news! We've Found the answer you've been seeking!
Question:
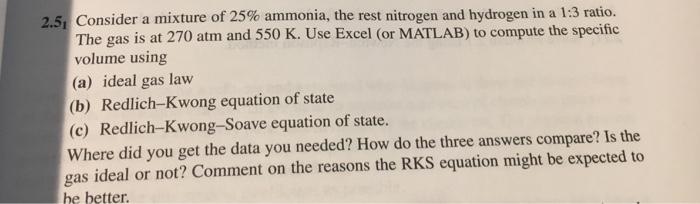
Transcribed Image Text:
2.5, Consider a mixture of 25% ammonia, the rest nitrogen and hydrogen in a 1:3 ratio. The gas is at 270 atm and 550 K. Use Excel (or MATLAB) to compute the specific volume using (a) ideal gas law (b) Redlich-Kwong equation of state (c) Redlich-Kwong-Soave equation of state. Where did you get the data you needed? How do the three answers compare? Is the gas ideal or not? Comment on the reasons the RKS equation might be expected to he better. 2.5, Consider a mixture of 25% ammonia, the rest nitrogen and hydrogen in a 1:3 ratio. The gas is at 270 atm and 550 K. Use Excel (or MATLAB) to compute the specific volume using (a) ideal gas law (b) Redlich-Kwong equation of state (c) Redlich-Kwong-Soave equation of state. Where did you get the data you needed? How do the three answers compare? Is the gas ideal or not? Comment on the reasons the RKS equation might be expected to he better.
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these mechanical engineering questions
-
You react nitrogen and hydrogen in a container to produce ammonia, NH3(g). The following figure depicts the contents of the container after the reaction is complete. a. Write a balanced chemical...
-
Consider a mixture of oxygen and nitrogen in the gas phase. How many independent properties are needed to fix the state of the system?
-
Consider a mixture of two gases A and B. Show that when the mass fractions mfA and mfB are known, the mole fractions can be determined from where MA and MB are the molar masses of A and B.
-
1. Consider the market for local rides (taxis, Uber Lyft, and so on), which is highly competitive. Suppose that the market is initially unregulated, but that the government imposes a binding price...
-
When a car drives off a cliff it rotates forward as it falls. For a higher speed off the cliff, will it rotate more, or less? (Consider the time that the unbalanced torque acts.)
-
For the following five sequences, what is the consensus sequence? 5GGGAGCG3 5GAGAGCG3 5GAGTGCG3 5GAGAACG3 5GAGAGCA3 a. 5GGGAGCG3 b. 5GAGAGCG3 c. 5GAGTGCG3 d. 5GAGAACG3
-
Bert C. Roberts Jr. was chairman of WorldComs board of directors. Immediately before that, he had been chairman of MCI, which WorldCom acquired on September 14, 1998, in a transaction valued at...
-
A stream of humid air containing 1.00 mole% H 2 O (v) and the balance dry air is to be humidified to a water content of 10.0 mole% H 2 O. For this purpose, liquid water is fed through a flow meter...
-
You have just been named Director of Data Administration of General Hardware Co. General Hardware maintains a large, central IS organization with several operational relational databases at its...
-
Assume that the Siletz River data are lognormally distributed. Find the following. (a) Peak flow of the 100-yr flood (b) Peak flow of the 50-yr flood (c) Probability that a flood will be less than or...
-
If cx = x, + x2 + 3x3 is minimized subject to x1 + x2 + x3 = 1, x2 0, show that two corners of the feasible set give the same minimum cx = 1. Find all solutions x, and explain geometrically how more...
-
What are the contents of an application record? Specify the Rule. And provide that an application record must contain the following information . Draft a notice of application for Tom Brad Pout, an...
-
I'm not going to buy from that store down the street, they don't have the quality of products that I want for my family. I will drive across the tracks (a different community) to buy want I want. "...
-
How do immunomodulatory cytokines orchestrate the delicate balance between pro-inflammatory and anti-inflammatory signaling pathways within the immune system, and what implications does this balance...
-
What kinds of inquiry approaches are best suited using each qualitative core design? Why? How does the nature of each university of houston core qualitative design differ from each other? What...
-
Why does Economists believe that the larger the amount individuals expect to save, the longer they will continue to search? Explain briefly this statment.
-
Need ads for any country and analysis for hofested culture of this country Through this ads. Provide a detailed answer.
-
Trade credit from suppliers is a very costly source of funds when discounts are lost. Explain why many firms rely on this source of funds to finance their temporary working capital.
-
A mixture of gases consists of 0.4 kg of oxygen, 0.7 kg of carbon dioxide, and 0.2 kg of helium. This mixture is maintained at 100 kPa and 27oC. Determine the apparent molecular weight of this...
-
A gas-turbine power plant operates on the simple Brayton cycle between the pressure limits of 100 and 800 kPa. Air enters the compressor at 30C and leaves at 330C at a mass flow rate of 200 kg/s. The...
-
Explain why the maximum flow rate per unit area for a given ideal gas depends only on P0 For an ideal gas with k = 1.4 and R = 0.287 kJ/kg·K, find the constant a such that m # /A* Po/VT
-
Consider the dynamic system, a mass, spring and damper structure, shown in Figure 2.2. (a) Draw a free-body diagram for the dynamic system (b) Derive the Input-Output model of the dynamic system k...
-
Two connected cars with an applied input force \(u(t)\) and negligible rolling friction can be represented by a translational mechanical system as shown below. (a) Draw the free-body diagrams of this...
-
Consider an RLC circuit consisting of a resistor \((R)\), an inductor \((L)\), and a capacitor \((C)\), connected in series, as depicted in Figure 2.4. Derive the Input-Output model of the network....

Study smarter with the SolutionInn App


