Consider a van der Waals gas: a) Calculate the isothermal compressibility. b) Calculate the expansion coefficient....
Fantastic news! We've Found the answer you've been seeking!
Question:
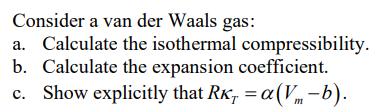
Transcribed Image Text:
Consider a van der Waals gas: a) Calculate the isothermal compressibility. b) Calculate the expansion coefficient. c) Show explicitly that RK, = alpha(Vm-b). Consider a van der Waals gas: a) Calculate the isothermal compressibility. b) Calculate the expansion coefficient. c) Show explicitly that RK, = alpha(Vm-b).
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the isothermal compressibility and the expansion coefficient of a van der Waals gas. Show, using Eulers chain relation, that T R = (V m b).
-
A van der Waals gas has a value of z = 1.00061 at 410. K and 1 bar and the Boyle temperature of the gas is 195 K. Because the density is low, you can calculate V m from the ideal gas law. Use this...
-
A sample consisting of 1.00 mol of a van der Waals gas is compressed from 20.0 dm 3 to 10.0 dm 3 at 300 K. In the process, 20.2 kJ of work is done on the gas. Given that = {(2a/RT) b}/C p,m, with C...
-
Problems 113 122. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for later sections, a final exam, or subsequent courses such as calculus. X...
-
Evan Schwartz has six jobs waiting to be processed through his machine. Processing time (in days) and due date information for each job are as follows: Sequence the jobs by FCFS, SPT, SLACK, and...
-
a. If I give you $30,000 today, and you invest it, how much money will you have in 25 years - assuming an interest rate of r? (Ignore inflation.) b. Suppose you win the lottery! Starting next year,...
-
Name the scale of measurement (nominal, ordinal, interval, ratio) for each of the following variables: a. One's age (in years) b. Size of soft drink (small, medium, large, extra large) c. Voting...
-
The Amberssen Specialty Company is a chain of 12 retail stores that sell a variety of imported gift items, gourmet chocolates, cheeses, and wines in the Toronto area. Amberssen has an IS staff of...
-
Please explain the following points in detail. The company name is Aritzia. 1. Market Concentration? a. What is the Market Concentration %? Show the numbers you used to calculate this (the sale of...
-
Tax Software Assignment - Fall 2023 Ms. Jeanette Letourneau (SIN 123-456-789) was born in Montreal on December 15, 1979. She has spent most of her working life a song writer but also has a part-time...
-
What are the Innovative Ways to Recover after a Failed Product Launch? Address the following: Definition of Product Failure and Recovery in regards to NPD Product Launch Understanding the Market...
-
Imagine a system that consists of only a single positively charged object. Is it possible to have in this system a closed surface that has a negative field line flux through it? (The charged object...
-
Suppose a certain planar surface has 2000 field lines passing through each square meter, normal to the surface. How many field lines pass through each square meter if the surface is tilted by...
-
Two parallel conducting plates carry equal and opposite charges. The plates are large relative to their separation distance, so we can assume the electric field between them is uniform. The potential...
-
A small charged ball is suspended at the center of a spherical balloon that is nestled snugly inside a cubical cardboard box. On one side, the balloon touches the wall of the box. (a) At this point...
-
Suppose a charged particle is located at the origin. What is the direction of the electric field at the point \((0.6,1.2)\) if the particle is \((a)\) positively charged and \((b)\) negatively...
-
Ayie decided to retired from the partnership on 1 September 20x7 due to illness. The goodwill is valued at RM120,000 on the retirement date. Goodwill account will not be maintained in the books. A...
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
Calculate the standard Gibbs energy of the reaction 4 HCl(g) + O2(g) 2 Cl 2 (g) + 2 H 2 O(l) at 298 K, from the standard entropies and enthalpies of formation given in the Data section.
-
The change in enthalpy is given by dH = C p dT + Vdp. The Clapeyron equation relates dp and dT at equilibrium, and so in combination the two equations can be used to find how the enthalpy changes...
-
Explain how metallic conductors and semiconductors are identified and explain their electrical and optical properties in terms of band theory.
-
A probability experiment consists of rolling a single fair die. (a) Identify the outcomes of the probability experiment. (b) Determine the sample space. (c) Define the event E = roll an even number....
-
Sophia has three tickets to a concert, but Yolanda, Michael, Kevin, and Marissa all want to go to the concert with her. To be fair, Sophia randomly selects the two people who can go with her. (a)...
-
A pair of fair dice is rolled. Fair die are die where each outcome is equally likely. (a) Compute the probability of rolling a seven. (b) Compute the probability of rolling snake eyes; that is,...

Study smarter with the SolutionInn App



