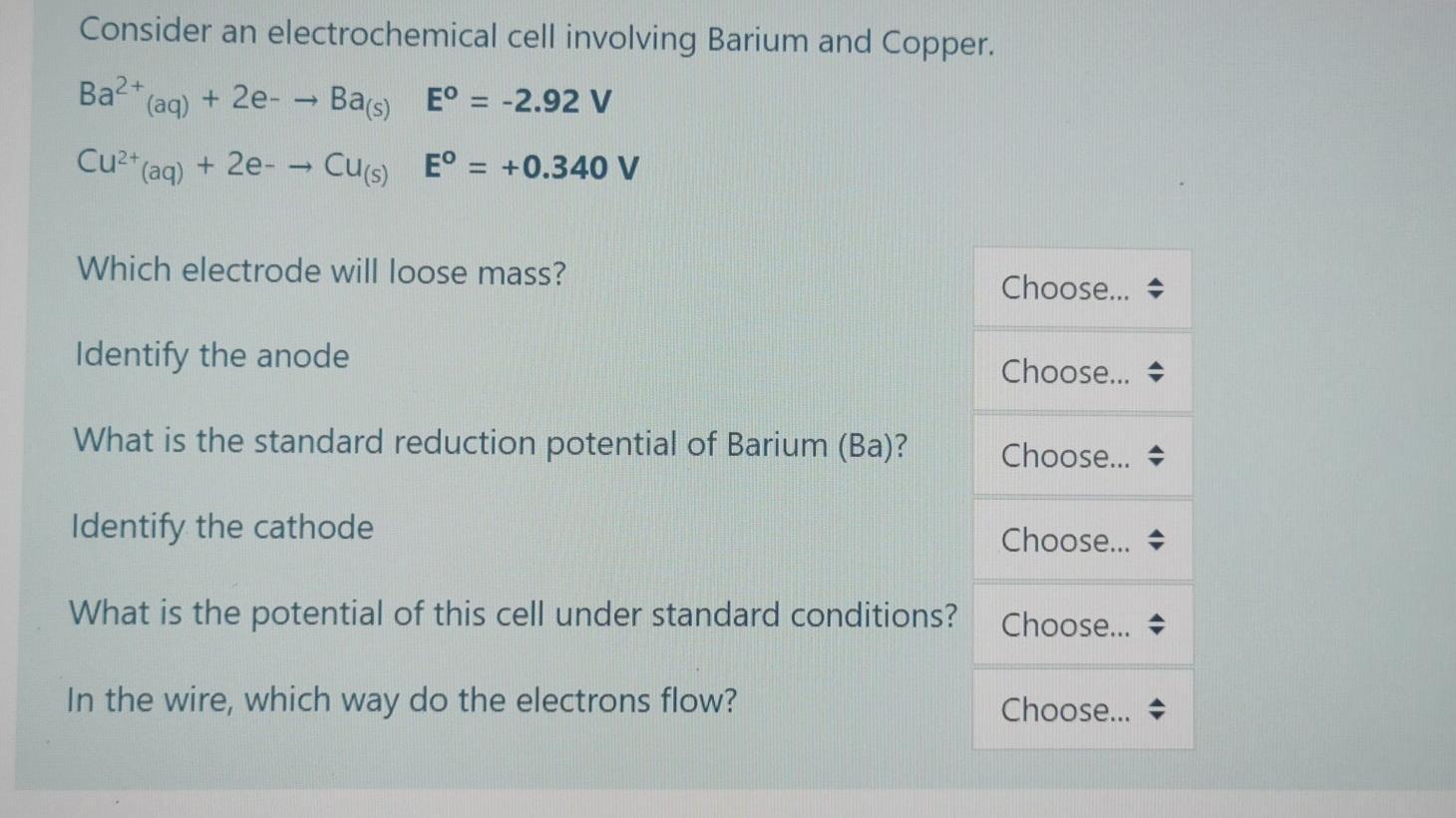
Consider an electrochemical cell involving Barium and Copper. Ba* (ag) + 2e- - Bas) E =...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider an electrochemical cell involving Barium and Copper. Ba* (ag) + 2e- - Bas) E° = -2.92 V Cu2" (aq) + 2e- – Cus) E° = +0.340 V %3D Which electrode will loose mass? Choose... + Identify the anode Choose... + What is the standard reduction potential of Barium (Ba)? Choose... + Identify the cathode Choose... + What is the potential of this cell under standard conditions? Choose... + In the wire, which way do the electrons flow? Choose... + Consider an electrochemical cell involving Barium and Copper. Ba* (ag) + 2e- - Bas) E° = -2.92 V Cu2" (aq) + 2e- – Cus) E° = +0.340 V %3D Which electrode will loose mass? Choose... + Identify the anode Choose... + What is the standard reduction potential of Barium (Ba)? Choose... + Identify the cathode Choose... + What is the potential of this cell under standard conditions? Choose... + In the wire, which way do the electrons flow? Choose... +
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
(a) What is the standard reduction potential of a standard hydrogen electrode? (b) Why is it impossible to measure the standard reduction potential of a single half-reaction?
-
What is the standard reduction potential for the oxidation of H2O (see Table 14-5)? Can this value be obtained from purple bacterial photosynthesis? Compare this to two-center photosynthesis.
-
Consider an electrochemical cell based on the reaction: 2H+(aq) + Sn(s) = Sn2+(aq) + H2(g). Which of the following actions would NOT change the measured cell potential? 1. Lowering the pH in the...
-
How many vector::push_back() operations are performed between lines 8 and 13 when this code is executed? The size of the input is the number of elements n in the vector v. Replace the question marks...
-
Comment on the following statement: Cash flow from operating activities is the most important section of the SCF. Hence, analysis should be focused on this section.
-
Three college students are considering operating a tutoring business in economics. This business would require that they give up their current jobs at the student recreation center, which pay...
-
Do the head-flowrate data shown in Fig. 12.7 appear to follow the similarity laws as expressed by Eqs. 12.39 and 12.40? Explain. Eq. 12.39 Eq. 12.40 (a) (b) Figure 12.7 (a) Open impeller, (b)...
-
Tony and Tina Roselli own and run TNTs Pizza Restaurant. Tony is responsible for managing the day-to-day aspects, hiring workers, and overseeing the kitchen, building, and grounds. He is the chief...
-
Provide documentation of a step by step training plan for a beta fish. What will be the primary reinforcer? How will you cue the behavior? What schedule of reminforcement will you use. What is the...
-
GUI Assignment You will be creating a seating arrangement application using JavaFX. The project will comprise of two sections: the interface and the Java code. The application will feature a 3x3...
-
Briefly Discuss the Introduction of COCA-COLA (MUST BE CATCHY) Environmental Analysis, SWOT Consumer Behavior, and the 4ps, parts of Add and Promotion, Social Media Add, Printed Add, and Conclusion.
-
Assets & Liabilities Provide a list of three business assets and three liabilities Assets Cost (over 12 months) Delivery van $30.000,00 Massage chairs $3000,00 Dcor $800,00 Total asset amount...
-
On May 20, White Repair Service extended an offer of $108,000 for land that had been priced for sale at $140,000. On May 30, White Repair Service accepted the seller's counteroffer of $115,000. On...
-
Vernon, Inc., has a 2 0 2 3 net Section 1 2 3 1 gain of $ 5 5 , 0 0 0 and had a $ 6 2 , 0 0 0 net Section 1 2 3 1 loss in 2 0 2 2 , For 2 0 2 3 , Vernon's net Section 1 2 3 1 gain is treated as:
-
Blossom Corporation purchased a limited-life intangible asset for $336000 on May 1, 2024. It has a useful life of 10 years. What total amount of amortization expense should have been recorded on the...
-
Based on the Harvard Business Review "Blockbuster Inc. & Technological Substitution (A): Achieving Dominance in the Video Rental Industry" case, (1) How does a revenue sharing contract work? Under...
-
A company will make the following annual lease lease i payments over the course of a 3-year" $150, $190 $160. Rent expense in the first year? (straight-line depreciation method) A company buys a $48...
-
In Exercises find dy/dx by implicit differentiation. xy - y = x
-
Blood contains several acidbase systems that tend to keep its pH constant at about 7.4. One of the most important buffer systems involves carbonic acid and hydrogen carbonate ion. What must be the...
-
Magnesium ammonium phosphate is an unusual salt in that it dissolves to form Mg2+, NH4+, and PO43 ions. Ksp for magnesium ammonium phosphate equals 2.5 1013. Calculate: (a) Its molar solubility; (b)...
-
Write the structure of propylbenzene. Write the structure of p-dichlorobenzene.
-
Find the probability that a randomly selected person has an IQ score higher than 125. Is this an unusual event? Explain. In a standardized IQ test, scores are normally distributed, with a mean score...
-
Find the probability that the number of U.S. adults who say they have had someone take over their email accounts without their permission is (a) at most 40, (b) less than 45, and (c) exactly 48....
-
A random sample of 60 people is selected from this population. What is the probability that the mean IQ score of the sample is greater than 105 ? Interpret the result. In a standardized IQ test,...

Study smarter with the SolutionInn App


