Consider the acid-base nature of barium nitrite, Ba(NO2)2, when it is dissolved in water. (1) What...
Fantastic news! We've Found the answer you've been seeking!
Question:

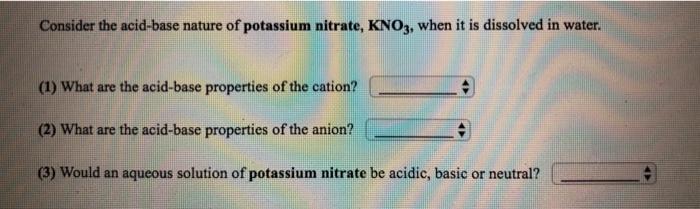
Transcribed Image Text:
Consider the acid-base nature of barium nitrite, Ba(NO2)2, when it is dissolved in water. (1) What are the acid-base properties of the cation? (2) What are the acid-base properties of the anion? (3) Would an aqueous solution of barium nitrite be acidic, basic or neutral? Consider the acid-base nature of potassium nitrate, KNO3, when it is dissolved in water. (1) What are the acid-base properties of the cation? (2) What are the acid-base properties of the anion? (3) Would an aqueous solution of potassium nitrate be acidic, basic or neutral? Consider the acid-base nature of barium nitrite, Ba(NO2)2, when it is dissolved in water. (1) What are the acid-base properties of the cation? (2) What are the acid-base properties of the anion? (3) Would an aqueous solution of barium nitrite be acidic, basic or neutral? Consider the acid-base nature of potassium nitrate, KNO3, when it is dissolved in water. (1) What are the acid-base properties of the cation? (2) What are the acid-base properties of the anion? (3) Would an aqueous solution of potassium nitrate be acidic, basic or neutral?
Expert Answer:
Answer rating: 100% (QA)
3 We have to determine acidBak nature of BacNO 2 f aqueous Bacaq 2 N000 a... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
When acetone is dissolved in water containing 18O instead of ordinary 16O (i.e., H2 18O instead of H2 16O), the acetone soon begins to acquire 18O and becomes The formation of this oxygen-labeled...
-
A certain acid HX is dissolved in water to make a 0.10M solution. At equilibrium, the concentration of X^- ions is 1.0 x 10^-5 M. The Pka of HX is approximately? A. 3 B. 9 C. 7 D. 12 E. 5
-
If 2 g NaOH is dissolved in water to make 250 ml solution . Give the molarity of the solution.
-
Q6) From the product_t table, return the number of rows of products with a natural ash finish OR a cherry finish Q7) From the customer_t table, return all entries with a postal code beginning with 9,...
-
Ten samples with five observations each have been taken from the Beautiful Shampoo Company plant in order to test for volume dispersion in the shampoo bottle-filling process. The average sample range...
-
What is meant by the term adequate disclosure, and how do accountants fulfill this requirement in the preparation of financial statements?
-
In the benzene adsorber of Example 9.7, the flow rate is increased to \(0.25 \mathrm{~m}^{3} / \mathrm{s}\). Calculate the breakthrough time and the fraction of the bed adsorption capacity that has...
-
Mifflin Corporation manufactures a single product. The standard cost per unit of product is shown below. Direct materials1 pound plastic at $7.00 per pound .... $ 7.00 Direct labor1.5 hours at $12.00...
-
Explain how a company uses a database management system to manage data collection, manipulate data and realize benefits from usage of a database management system.?
-
We want to design a tension spring with a spring outer diameter of 32 mm. The range of tensile load is 110 160 N, and the deformation range is 140~ 170 mm. We are going to use SWO-B as the material....
-
A centrifuge slows down from 691 rpm to 281 rpm in 14 seconds. Assuming constant angular acceleration, determine the number of revolutions it goes through in this time. Express your answer using zero...
-
Merrick Industries Ltd . manufactures computer keyboards. Each keyboard goes through two different production departments.Department A applies overhead based on machine hours while Department B...
-
1. If a certain spring is stretched 2 m, it will produce a force of 10 N. Set up an integral to find the amount of work done in stretching the spring an additional 3 m.
-
Robert s Painting Service Inc. Contracted a $ 9 0 , 0 0 0 painting job on Nov. 1 5 th . The customer paid $ 4 5 , 0 0 0 when the contract was signed. Robert recorded this money as Unearned Painting...
-
Test the following polar equations for symmetry with respect to the line 0 = /2, the polar axis and the pole. A. r = -3sin(20) B. r2 1 2cos(0) Remember: cos(-0) = cos(0) sin(-0) = -sin(0) Write the...
-
The year end for a sole trader is 3 0 November 2 0 X 7 . The business pays for its gas by a standing order of 2 0 0 per month. On 1 December 2 0 X 6 , the statement from the gas supplier showed that...
-
Once a mortgage application is received by the mortgage provider, and before proceeding with its assessment, the application itself needs to be checked for completeness. If the application is...
-
In Problem use geometric formulas to find the unsigned area between the graph of y = f(x) and the x axis over the indicated interval. f(x) = x + 5; [0, 4]
-
What do the bond angles of boron trifluoride suggest about the hybridization state of the boron atom?
-
When limonene (Section 23.3) is heated strongly, it yields 2 mol of isoprene. What kind of reaction is involved here?
-
Write the resonance structure that would result from moving the electrons as the curved arrows indicate. Be sure to include formal charges if needed. (a) (b) (c) (d) 079 HIN:
-
True or False: Annual worth analysis is the most popular DCF measure of economic worth.
-
Consider a palletizer at a bottling plant that has a first cost of \($150,000,\) operating and maintenance costs of \($17,500\) per year, and an estimated net salvage value of \($25,000\) at the end...
-
True or False: Unless non-monetary considerations dictate otherwise, choose the mutually exclusive investment alternative that has the greatest annual worth over the planning horizon.

Study smarter with the SolutionInn App


