Question: Consider the beta decay 198 198 Au Hg+B+v where Hg represents a mercury nucleus in an excited state at energy 1088 MeV above the
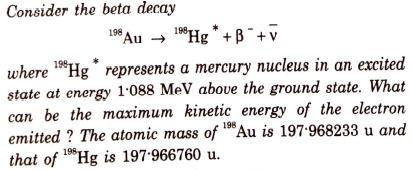
Consider the beta decay 198 198 Au Hg+B+v where Hg represents a mercury nucleus in an excited state at energy 1088 MeV above the ground state. What can be the maximum kinetic energy of the electron emitted? The atomic mass of 198 Au is 197-968233 u and that of Hg is 197.966760 u.
Step by Step Solution
3.29 Rating (167 Votes )
There are 3 Steps involved in it
The detailed ... View full answer

Get step-by-step solutions from verified subject matter experts


