Consider the dissolution of CaCl2: An 11.0-g sample of CaCl2 is dissolved in 125g of water, withboth
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider the dissolution of CaCl2:
An 11.0-g sample of CaCl2 is dissolved in 125g of water, withboth substances at 25.0 degrees Celsius. Calculate the finaltemperature of the solution assuming no heat lost to thesurroundings and assume the solution has a specific heat capacityof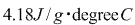 .
.
Related Book For 

Posted Date:




