Consider the endothermic reaction 2H2S(g) + SO2 (g) 3S(s) + 2HO(g) at equilibrium. Match each change...
Fantastic news! We've Found the answer you've been seeking!
Question:
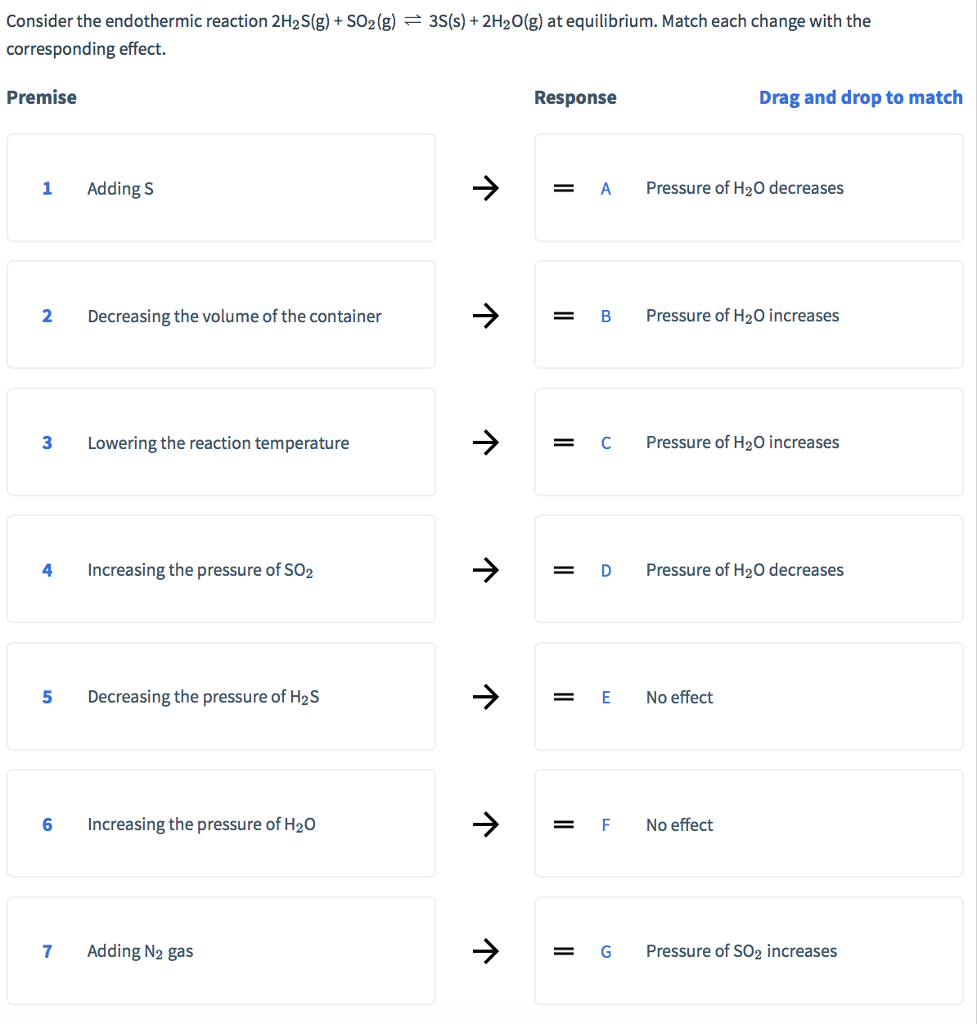
Transcribed Image Text:
Consider the endothermic reaction 2H2S(g) + SO2 (g) 3S(s) + 2H₂O(g) at equilibrium. Match each change with the corresponding effect. Premise 1 2 3 4 5 6 7 Adding S Decreasing the volume of the container Lowering the reaction temperature Increasing the pressure of SO2 Decreasing the pressure of H₂S Increasing the pressure of H₂O Adding N₂ gas 个 ↑ 个 ↑ ↑ Response = A = B = = C = D = E F = G Pressure of H₂O decreases Drag and drop to match Pressure of H₂O increases Pressure of H₂O increases Pressure of H₂O decreases No effect No effect Pressure of SO2 increases Consider the endothermic reaction 2H2S(g) + SO2 (g) 3S(s) + 2H₂O(g) at equilibrium. Match each change with the corresponding effect. Premise 1 2 3 4 5 6 7 Adding S Decreasing the volume of the container Lowering the reaction temperature Increasing the pressure of SO2 Decreasing the pressure of H₂S Increasing the pressure of H₂O Adding N₂ gas 个 ↑ 个 ↑ ↑ Response = A = B = = C = D = E F = G Pressure of H₂O decreases Drag and drop to match Pressure of H₂O increases Pressure of H₂O increases Pressure of H₂O decreases No effect No effect Pressure of SO2 increases
Expert Answer:

Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these chemistry questions
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
Consider the reaction E + F G + H, which has the following reaction coordinate diagram. Which one of the following statements is true? a. The activation energy is greatest for the forward reaction....
-
Consider the hypothetical reaction B E + F which is assumed to occur by the mechanism Where B* represents a B molecule with enough energy to surmount the reaction energy bar70. Consider the following...
-
It is an extremely hot and sunny day on Far Rockaway beach. However, there is only one shop selling non-dairy ice cream; the shop is a monopolist retailer of ice cream. There are two types of people...
-
LeAnn Bryant's check book lists the following: The November bank statement shows *This is the correct amount for check number 624. Required Prepare Bryant's bank reconciliation at November 30, 20X6....
-
Obvert the following propositions and state whether the obverse is logically equivalent or not logically equivalent to the given proposition. a. All radically egalitarian societies are societies that...
-
The defendant, Sterile Technologies, Inc., purchased a sterilizer from the plaintiff, Troy Boiler Works, on an installment payment plan. The defendant was to make installment payments charged with
-
Premier Bank and Trust is considering giving Alou Company a loan. Before doing so, management decides that further discussions with Alou's accountant may be desirable. One area of particular concern...
-
According to the Civil Code of the Philippines, Article 1 7 6 7 defines partnership as: By the contract of partnership two or more persons bind themselves to contribute money, property, or industry...
-
Ellipses Corp is a small business that operates in Herndon, VA. The company is located at10 Period Lane, Herndon, VA 20170. Its federal Employer Identification Number is 77-7777777, and its...
-
This project will have two classes: A test class and a Bank Account class. Construct the Bank Account class by pasting the following code into BlueJ: public class BankAccount { public BankAccount...
-
The change in 4.17.5 requires many existing LW/SW instructions to be converted into two-instruction sequences. If this is needed for 50% of these instructions, what is the overall speedup achieved by...
-
Penelope Pundit, an economics reporter, states that the European Union (EU) is increasing its productivity very rapidly in all industries. She claims that this productivity advance is so rapid that...
-
In your state the minimum wage is $12 per hour. Yet, you notice that many fast-food restaurants have posted help wanted signs for jobs paying $15 per hour. What does this tell you about the minimum...
-
Two young entrepreneurs, Garrett Camp and Travis Kalanick, created Uber in 2009 to alleviate a common frustration: how to find a taxi when you need to get somewhere and there arent any taxis...
-
Oregon recently became the first state to adopt legislation capping housing rents statewide. Explain how Oregons policy will affect landlords, renters, and the quality of rental units available.
-
What is constitutional law in the U.S? Where did it originate? By what right are laws made? Who interprets laws? How has seminal constitutional and contractual case law and the influence of these on...
-
Comptech Ltd is a manufacturer of optical equipment. In September 2019, Ed Thompson the Chief Research Officer, attended a conference in Switzerland that focused on optical developments for the 21st...
-
Calculate the higher heating value (HHV) of methane (CH4) per unit mass of fuel at (a) 298 K, and (b) 500 K.
-
A steam heating system for a building 175 m high is supplied from a boiler 20 m below ground level. Dry, saturated steam is supplied from the boiler at 300 kPa, which reaches the top of the building...
-
An air conditioning system operates at a total pressure of 1 atm consists of a heating section and humidifier which supplies wet steam (saturated water vapor) at 1 atm. Air enters the heating section...
-
How can speakers use anxiety to their advantage?
-
What are common psychological, physical, and behavioral reactions associated with speech anxiety?
-
Describe strategies for making people the focus of your presentations.

Study smarter with the SolutionInn App


