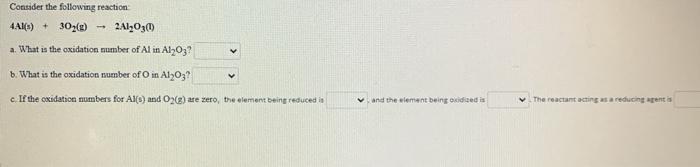
Consider the following reaction: 4.Al(s) + 302(g) 1 2AlO3(1) a. What is the oxidation number of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following reaction: 4.Al(s) + 302(g) 1 2Al₂O3(1) a. What is the oxidation number of Al in Al2O3? b. What is the oxidation number of O in Al₂O37 c. If the oxidation numbers for Al(s) and O2(g) are zero, the element being reduced is and the element being oxidized is The reactant acting as a reducing agent is Consider the following reaction: 4.Al(s) + 302(g) 1 2Al₂O3(1) a. What is the oxidation number of Al in Al2O3? b. What is the oxidation number of O in Al₂O37 c. If the oxidation numbers for Al(s) and O2(g) are zero, the element being reduced is and the element being oxidized is The reactant acting as a reducing agent is
Expert Answer:
Answer rating: 100% (QA)
Sol 4 Als 3 O g 2 AlO3 1 a Oxidation number of oxygen is generally t... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
The accountant for a subunit of Mountain Sports Company went on vacation before completing the subunits monthly performance report. This is as far as she got: Requirements 1. Complete the performance...
-
Accounts receivable analysis The following data are taken from the financial statements of Saladin Inc. Terms of all sales are 2/10, n/60 A. Determine for each year (1) The accounts receivable...
-
In Hollywood movies, it is common to have product placement (having products from sponsored companies, such as cars, appear in movies without telling viewers that these are commercials). As a...
-
Inland Equipment uses the allowance method to account for uncollectible accounts. On December 31,2010, Allowance for Uncollectible Accounts has a $600 credit balance. Journalize the year-end...
-
Og is the leader of the surprisingly mathematically advanced, though technologically run-of-the-mill, Calm Waters caveman tribe. He must decide on the number of stone clubs and stone axes to be...
-
1) Explain how the process of valuing a high-growth company differs from valuing an established company. 2) How does the total market for a new product differ from a company's addressable market?...
-
This problem continues the Draper Consulting, Inc., situation from Problem 2-62 of Chapter 2. Start from the trial balance and the posted T-accounts that Draper Consulting, Inc., prepared at December...
-
Draft a Multinational Integration Strategic Human Resources Plan. You may select either a 'real' company or a 'fictitious' company. Your company should be in the industry vertical. You are to lead...
-
In the movie, 12 Angry Men, the jurors wanted to say the defendant was guilty so they could get on with their lives. In the beginning of the movie, the audience doesn't know anything about the case's...
-
Two children, Tommy aged three and Shea a one-year-old, recently arrived at your service with their grandmother, Carla. Carla lives in New Zealand but came to Australia to help her daughter Bea with...
-
How do the linear thermal expansion coefficients for steel, copper, and aluminum compare? Fundamentally, what are the differences occurring at the atomic level to cause this difference?
-
Suppose you want to observe the molecular gas in a galaxy with redshift z using the rotational transition of CO J=4-3. What frequency would you observe this transition at?
-
ANSWER WITHIN 30 MIN Cyral and Amira are cousins. Amira is a soware engineer and Cyral is in construcon management. They have shared a passion for geng involved in innovave projects. In 2021, Amira...
-
Explain why it is important for team members and stakeholders need to be aware of quality requirements? Why are quality requirements and metrics effective tools to use when measuring performance?...
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
The following electron configurations represent excited states. Identify the element, and write its ground-state condensed electron configuration. (a) 1s2 2s2 3p2 4p1, (b) [Ar] 3d10 4s1 4p4 5s1, (c)...
-
Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23oC. (a) What mass of O2 does the tank...
-
KCl has the same structure as NaCl. The length of the unit cell is 6.28 . The density of KCl is 1.984 g/cm3, and its formula mass is 74.55 amu. Using this information, calculate Avogadro's number?
-
A cylindrical tank of methanol has a mass of \(60 \mathrm{~kg}\) and a volume of \(75 \mathrm{~L}\). Determine the methanol's weight, density, and specific gravity. Take the gravitational...
-
A mass of \(0.5-\mathrm{kg}\) of argon is maintained at \(1400 \mathrm{kPa}\) and \(40^{\circ} \mathrm{C}\) in a tank. What is the volume of the tank?
-
The air in an automobile tire with a volume of \(0.015 \mathrm{~m}^{3}\) is at \(30^{\circ} \mathrm{C}\) and \(140 \mathrm{kPa}\) (gage). Determine the amount of air that must be added to raise the...

Study smarter with the SolutionInn App


