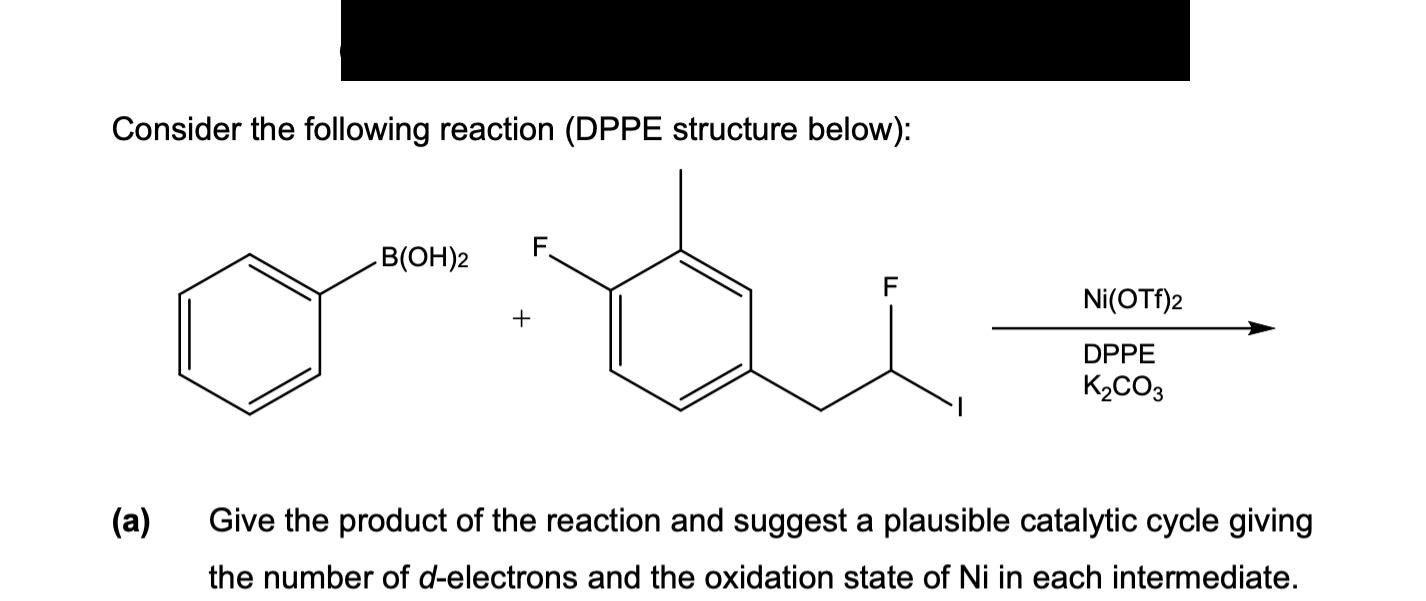
Consider the following reaction (DPPE structure below): F. B(OH)2 F Ni(OTf)2 + DPPE K2CO3 () Give...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following reaction (DPPE structure below): F. B(OH)2 F Ni(OTf)2 + DPPE K2CO3 (а) Give the product of the reaction and suggest a plausible catalytic cycle giving the number of d-electrons and the oxidation state of Ni in each intermediate. Consider the following reaction (DPPE structure below): F. B(OH)2 F Ni(OTf)2 + DPPE K2CO3 (а) Give the product of the reaction and suggest a plausible catalytic cycle giving the number of d-electrons and the oxidation state of Ni in each intermediate.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Burke Fabricators completed two jobs in June. Burke Fabricators recorded the following costs assigned to the jobs by the companys activity-based costing system: Job 622 required 2,400 parts, 77,500...
-
There are two major cell phone providers in the Colorado Springs, Colorado, area, one called HTC and the other, Mountain Communications. We want to investigate whether there is a difference in the...
-
A former employee brought an action against his former employer alleging unlawful retaliation in violation of Title VII of the Civil Rights Act of 1964 and wrongful discharge contrary to Wisconsin...
-
Which of the following procedures is most telling in addressing the existence assertion for accounts payable? a. Test for unrecorded liabilities. b. Confirm balances with creditors. c. Verify the...
-
Raptor Fuels produces three grades of gasoline Regular, Premium, and Super. All of these are produced by blending two types of crude oil Crude A and Crude B. The two types of crude contain specific...
-
How do individual differences in personality, values, and attitudes influence team dynamics and organizational culture ?
-
Consider a scenario in which state colleges must actively recruit students. California Sci has $750,000 in assets available. Its Board of Regents has to consider several options. The board may decide...
-
1 ) Create a balance sheet for a company of your choosing. 2 ) Your balance sheet must contain the following: ( a ) 5 current assets ( b ) 4 fixed assets ( c ) 3 current liabilities ( d ) 2 non -...
-
Professional Research Hincapie Co. manufactures specialty bike accessories. The company is most well known for its product quality, and it has offered one of the best warranties in the industry on...
-
Cargo Company purchased land from Hazard Company for 20,000,000 on September 13, 2015. On October 31, 2015, it learned that the site was contaminated. As a result, Cargo Company sued Hazard Company...
-
Using the information from BE19-2, assume this is the only difference between Oxfords pretax financial income and taxable income. Prepare the journal entry to record the income tax expense, deferred...
-
Sujo Corporation commenced operations in early 2015. The corporation incurred HK\($60\),000,000 of costs such as fees to underwriters, legal fees, governmental fees, and promotional expenditures...
-
YellowCard Company manufactures accessories for iPods. It had the following selected transactions during 2015. (For any part of this problem requiring an interest or discount rate, use 10%.) 1....
-
In order for a price ceiling to bind, it a.must be set above the equilibrium price, and will likely cause a shortage. b.must be set below the equilibrium price, and will likely cause a shortage. ...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
Compare and contrast the structures of the following solids. a. CO2(s) and H2O(s) b. NaCl(s) and CsCl(s)
-
How would transition metal ions be classified using the Lewis definition of acids and bases? What must a ligand have to bond to a metal? What do we mean when we say that a bond is a coordinate...
-
A sample consisting of 22.7 g of a nongaseous, unstable compound X is placed inside a metal cylinder with a radius of 8.00 cm, and a piston is carefully placed on the surface of the compound so that,...
-
According to the monetary approach to the balance of payments, what type of economic policies would help a country resolve a balance of trade deficit?
-
We discussed risk aversion as being descriptive of investor behavior. Can Do you think of any real-world behavior that you might consider to be evidence of the existence of risk preferrers?
-
Pick three developing countries and create a country risk index for them. Rank them ordinally in terms of factors that you can observe (exports, GDP growth, reserves, etc.) by looking at...

Study smarter with the SolutionInn App



