(5) A wave with = 10 s and k = 7 + 6- 3k m has...
Fantastic news! We've Found the answer you've been seeking!
Question:
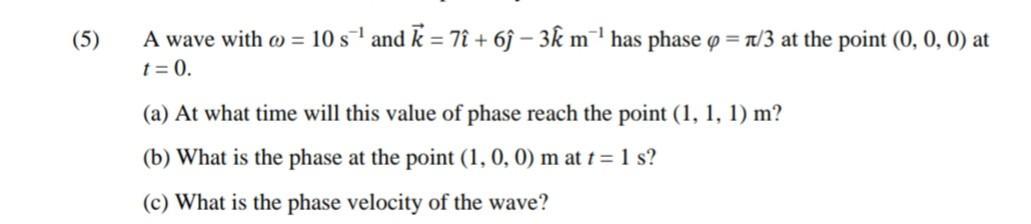
Transcribed Image Text:
(5) A wave with = 10 s¹ and k = 7î + 6ĵ- 3k m¹ has phase = /3 at the point (0, 0, 0) at t = 0. (a) At what time will this value of phase reach the point (1, 1, 1) m? (b) What is the phase at the point (1, 0, 0) m at t = 1 s? (c) What is the phase velocity of the wave? (5) A wave with = 10 s¹ and k = 7î + 6ĵ- 3k m¹ has phase = /3 at the point (0, 0, 0) at t = 0. (a) At what time will this value of phase reach the point (1, 1, 1) m? (b) What is the phase at the point (1, 0, 0) m at t = 1 s? (c) What is the phase velocity of the wave?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
Consider the following system at equilibrium at 25oC: PCl3(g) + Cl2(g) PCl5(g) Go = 92.50 kJ What will happen to the ratio of partial pressure of PCl5 to partial pressure of PCl3 if the temperature...
-
The following system of equations is designed to determine concentration (the cs in g/m 3 ) in a series of coupled reactors as a function of the amount of mass input to each reactor (the right-hand...
-
Solve the following system of equations and check your solution. 4x + y = -13 x - 5y = -19
-
Ava bought a house on 1 July 2000 for 62,000. She occupied the entire property as her PPR until 1 August 2007 when she began using one-quarter of the house for business purposes. This continued until...
-
You work for a firm producing fitness equipment. You have been told that the demand curve for the firms main producta multistation home gymis linear. You have been provided with price and quantity...
-
Primare Corporation provided the following data for last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor Manufacturing overhead applied...
-
The chemical potential of a pure substance is equal to the (a) Specific Gibbs' free energy (b) Molar entropy (c) The Gibbs' free energy (d) Molar Gibbs' free energy.
-
Hilburn Manufacturing Corporation had the following transactions for its job order costing operation. Prepare general journal entries to record these transactions. Jan. 1 Purchased materials on...
-
Raymond B. Cattell was a psychologist who utilized thestatistical technique of factor analysis to determine underlyingpersonality traits. His definition of personality was very concise:personality is...
-
You are meeting with executives of Cooper Cosmetics Corporation to arrange your firms engagement to audit the corporations financial statements for the year ending December 31. One executive suggests...
-
On January 1 of the current year, CCH Corporation entered into the following lease contract. Based on the facts, CCH Corporation classifies the lease as an operating lease. The company has a 5% cost...
-
Use the data set, which represents the ages of 30 executives. Which ages are above the 25th percentile? 43 57 65 47 57 41 56 53 61 54 56 50 66 56 50 61 47 40 50 43 54 41 48 45 28 35 38 43 42 44
-
Value-added refers to the difference in the overall value of what and what?
-
Consider a shipping company that carries freight by truck. Name three inputs to this operation.
-
Identify several situations in which more than one of the following would be practiced simultaneously: production management, project management, event management, and incident management.
-
The distribution of the ages of the winners of the Tour de France from 1903 to 2016 is approximately bell-shaped. The mean age is 27.9 years, with a standard deviation of 3.3 years. Use the...
-
3. List the following types of electromagnetic radiation in order of increasing frequency, increasing wavelength, increasing energy. c) microwave radiation a) gamma rays b) infrared radiation _+) -)...
-
Wimot Trucking Corporation uses the units-of-production depreciation method because units-of-production best measures wear and tear on the trucks. Consider these facts about one Mack truck in the...
-
At 2200oC, K = 0.050 for the reaction N2(g) + O2(g) 2NO(g) What is the partial pressure of NO at equilibrium assuming the N2 and O2 had initial pressures of 0.80 atm and 0.20 atm, respectively?
-
Consider the following reactions at some temperature: 2NOCl( g) 2NO( g) + Cl2( g) K = 1.6 1025 2NO( g) N2( g) + O2( g) K = 1 1031 For each reaction some quantities of the reactants were placed in...
-
Use the relative ionic radii in Fig. 13.8 to predict the structures expected for CsBr and KF. Do these predictions agree with observed structures? The ionic radius of Cs+ is 169 pm. Figure 13.8 Lit...
-
Apart from the needs of children, do you think the state has a legitimate interest in encouraging marital permanency through its divorce laws? If so, how should the state's interest be balanced with...
-
The record supports the following facts. The petitioner filed for divorce from the respondent on grounds of irreconcilable differences. He subsequently moved to amend the petition to assert the fault...
-
What is the best interest standard?

Study smarter with the SolutionInn App



