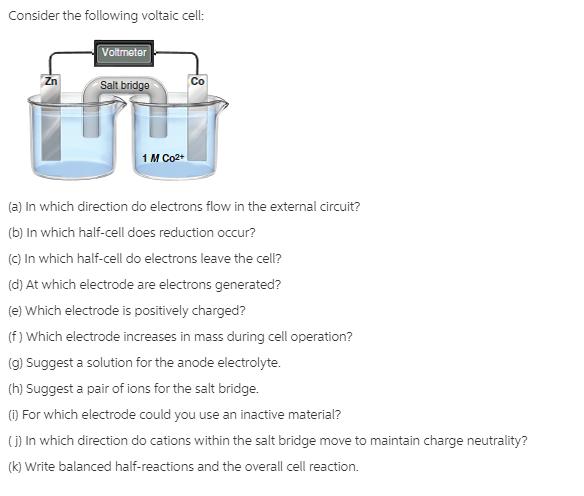
Consider the following voltaic cell: Voltmeter Salt bridgo Zn Co 1 M Co2+ (a) in which...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following voltaic cell: Voltmeter Salt bridgo Zn Co 1 M Co2+ (a) in which direction do electrons flow in the external circuit? (b) In which half-cell does reduction occur? (C) In which half-cell do electrons leave the cell? (d) At which electrode are electrons generated? (e) Which electrode is positively charged? (f ) Which electrode increases in mass during cell operation? (g) Suggest a solution for the anode electrolyte. (h) Suggest a pair of ions for the salt bridge. () For which electrode could you use an inactive material? ) In which direction do cations within the salt bridge move to maintain charge neutrality? (k) Write balanced half-reactions and the overall cell reaction. Consider the following voltaic cell: Voltmeter Salt bridgo Zn Co 1 M Co2+ (a) in which direction do electrons flow in the external circuit? (b) In which half-cell does reduction occur? (C) In which half-cell do electrons leave the cell? (d) At which electrode are electrons generated? (e) Which electrode is positively charged? (f ) Which electrode increases in mass during cell operation? (g) Suggest a solution for the anode electrolyte. (h) Suggest a pair of ions for the salt bridge. () For which electrode could you use an inactive material? ) In which direction do cations within the salt bridge move to maintain charge neutrality? (k) Write balanced half-reactions and the overall cell reaction.
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Consider the following voltaic cell: (a) Which electrode is the cathode? (b) How would you determine the standard emf generated by this cell? (c) What is the change in the cell voltage when the ion...
-
What is the cell potential of the following voltaic cell at 25oC? Zn(s)|Zn2+(0.200 M)||Ag + (0.00200 M)|Ag(s)
-
Write the overall cell reaction for the following voltaic cell. Fe(s) |Fe2+(aq)||Ag + (aq)|Ag(s)
-
An astronaut must journey to a distant planet, which is 200 light-years from Earth. What speed will be necessary if the astronaut wishes to age only 10 years during the round trip?
-
Long-term complications of diabetes include a. Blindness. b. Kidney disease. c. Circulatory disorders. d. All of these are correct. e. None of these is correct.
-
For a test of H 0 : = 0 against H a : > 0 based on n = 30 observations and using a = 0.05 significance level, P(Type II error) = 0.36 at = 4. Identify the response that is incorrect. a. At = 5,...
-
Almetals, Inc., a Michigan company, entered into a contract with the German firm Wickeder Westfalenstahl regarding the purchase of clad metal, a specialty metal used in a variety of industries but...
-
Scully Corporations comparative balance sheets are presented below. Additional information: 1. Net income was $22,630. Dividends declared and paid were $19,500. 2. All other changes in noncurrent...
-
Ethel has an existing loan which she wishes to refinance to obtain a lower rate. Her home has appraised for 352,000. She has requested a new loan in the amount of 264,000 to pay off her old loan and...
-
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Customer ID 652-54310 Results of A/R Confirmations Complete the worksheet below by reviewing the accounts receivable confirmation...
-
One characteristic of programming languages that varies widely from language to language is how parameters are passed. Among ALGOL, Pascal, Ada, C, C++, Java, and C#, no two languages pass parameters...
-
Theodor Adorno and Henry Jenkins come to different conclusions about how audiences interact with and are affected by popular culture. Using their ideas and examples from the documentary Trekkies 2,...
-
what you believe to be the major problem. In your response to this discussion board, type the problem you will address and describe what kind of problem it is: management, HR, etc. Then pose 3...
-
On Wednesday, Tom, a vintage car dealer, placed an advertisement in a weekly motor sports magazine offering to sell a Triumph TR6 for 10,000, cheque accepted. Chris saw the advertisement on Thursday...
-
In doing a five-year analysis of future dividends, the Dawson Corporation is considering the following two plans. The values represent dividends per share. Use Appendix B for an approximate answer...
-
Your parents live in a hotspot for COVID 19 where case counts are surging, but they only listen to news that puts a positive spin on the pandemic. What kind of listening are they employing?
-
The network diagram below shows four routers named Toby, Joan, Mohan and Boon. Assume that all the networks shown are aware of each other and have fully updated routing tables and router Boon was...
-
Find the radius of convergence in two ways: (a) Directly by the CauchyHadamard formula in Sec. 15.2. (b) From a series of simpler terms by using Theorem 3 or Theorem 4.
-
Nuclear scientists have synthesized approximately 1600 nuclei not known in nature. More might be discovered with heavyion bombardment using high-energy particle accelerators. Complete and balance the...
-
Acetonitrile (CH3CN) is a polar organic solvent that dissolves a wide range of solutes, including many salts. The density of a 1.80 M LiBr solution in acetonitrile is 0.826 g/cm3. Calculate the...
-
Two Fe(II) complexes are both low spin but have different ligands. A solution of one is green and a solution of the other is red. Which solution is likely to contain the complex that has the...
-
What are the implications of behavioral finance?
-
Suppose that you are a trader at the stock market. T-Mobiles stocks currently trade at $45 and the expected return is 9%. You have information that leads you to believe that by the end of year the...
-
You are considering purchasing a 10-year bond and follow the theory of rational expectations. If you have just read the annual report of the central bank in your country that states interest rates...

Study smarter with the SolutionInn App


