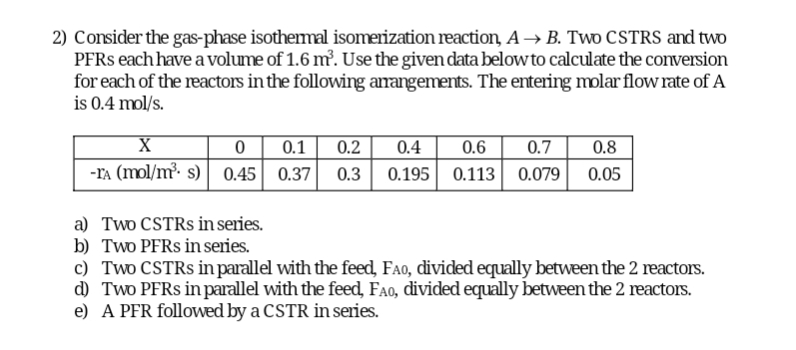
Question: Consider the gas - phase isothermal isomerization reaction, A B . Two CSTRS and two PFRs each have a volume of 1 . 6 m
Consider the gasphase isothermal isomerization reaction, Two CSTRS and two PFRs each have a volume of Use the given data below to calculate the conversion for each of the reactors in the following amangements. The entering molar flow rate of is
table
a Two CSTRs in series.
b Two PFRs in series.
c Two CSTRs in parallel with the feed, divided equally between the reactors.
d Two PFRs in parallel with the feed, divided equally between the reactors.
e A PFR followed by a CSTR in series.
Step by Step Solution
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock


Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


