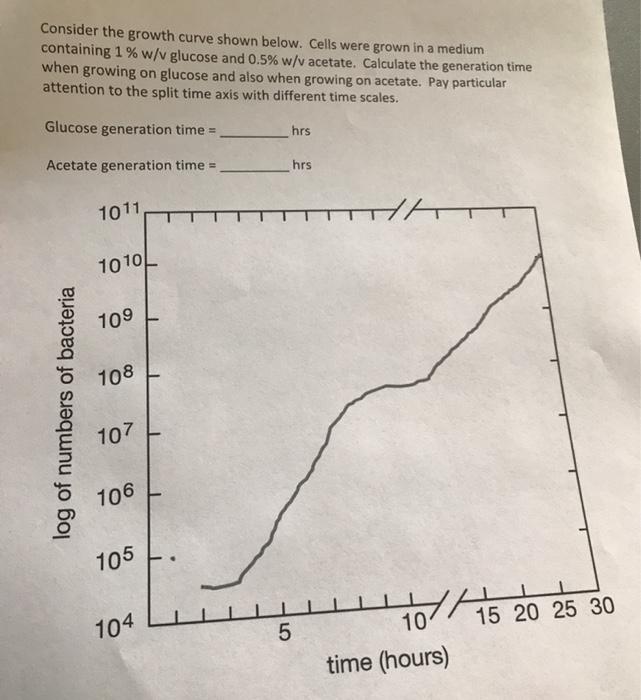
Consider the growth curve shown below. Cells were grown in a medium containing 1 % w/v...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the growth curve shown below. Cells were grown in a medium containing 1 % w/v glucose and 0.5% w/v acetate. Calculate the generation time when growing on glucose and also when growing on acetate. Pay particular attention to the split time axis with different time scales. Glucose generation time = hrs Acetate generation time = hrs 1011 1010 109 108 107 106 105 104 10 15 20 25 30 time (hours) HA log of numbers of bacteria LO Consider the growth curve shown below. Cells were grown in a medium containing 1 % w/v glucose and 0.5% w/v acetate. Calculate the generation time when growing on glucose and also when growing on acetate. Pay particular attention to the split time axis with different time scales. Glucose generation time = hrs Acetate generation time = hrs 1011 1010 109 108 107 106 105 104 10 15 20 25 30 time (hours) HA log of numbers of bacteria LO
Expert Answer:
Answer rating: 100% (QA)
The process and calculations step by step Number of generation in a given time N log N ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
If squalene were synthesized in a medium containing acetate whose carbonyl carbon were labeled with radioactive 14C which carbons in squalene would be labeled?
-
Find the diameter of the shaft required to transmit 60 kW at 150 rpm if the maximum torque exceeds 25% of the mean torque for a maximum permissible shear stress of 60 MN/mm. Find also the angle of...
-
Perform a SWOT analysis for Amazon.com. Pay particular attention to industry convergence. What are the strategic implications for Amazon?
-
ABC Corporation earned $300,000 in profit in the current tax year, which is significantly higher than its previous year's earnings. Thus, its shareholders expected large dividends in the current...
-
In the previous problem, what is the power delivered by the motor at the operating point in units of (a) Watts and (b) Horsepower? Previous problem A DC servomotor has a torque constant = 0.088 N-m/A...
-
Suppose that in six months we will need 500 ounces of gold, and that the current (time \(t=0\) ) forward price for delivery in 0.5 years (six months) is \[F\left(\begin{array}{lll}0 & 0 &...
-
The following data come from a study in which random samples of the employees of three government agencies were asked questions about their pension plan: Use the 0.01 level of significance to test...
-
Megan has her home and personal property insured under an unendorsed Homeowners 3 (special form) policy. Indicate whether each of the following losses is covered. If the loss is not covered, explain...
-
A:Write a research proposal based on one of the following topics: 1: Social Media is positively or negatively affecting the way we communicate. 2: Cellphones should or should not be allowed in the...
-
Lou Barlow, a divisional manager for Sage Company, has an opportunity to manufacture and sell one of two new products for a five- year period. His annual pay raises are determined by his division's...
-
The main objective of the financial statements is to provide useful information to (a) government in deciding if the company is respecting tax laws. (b) increase the value of the company. (c)...
-
Pricing and Production Decisions PoolVac, Inc. manufactures and sells a single product called the "Sting Ray," which is a patent-protected automatic cleaning device for swimming pools. PoolVac's...
-
The management of OBL Inc., a consulting company, had a meeting to discuss the potential impact of new social movements on their core business. The board of directors agreed that to avoid potential...
-
Divide: Express your answer in lowest terms. 7 37 12
-
Total revenues $69,495 Net operating assets (NOA) $21,128 Net operating profit after tax (NOPAT) $3,503 Net operating profit margin (NOPM) 4.8% Net operating asset turnover (NOAT) 3.29 Discount rate...
-
we want to study the quality of a noisy communication channel of a digital camera. for this, we consider that the source delivered the colors is modeled by the following transition matrix: P [P(Y/X)]...
-
The seller of investment in equity securities, received P556,640 as net proceeds from sale of its investment in equity securities, net of 6/10 of 1% of selling price. How much is the corresponding...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Give the product of the following reaction: If the terminal sp2 carbon of the substituent bonded to the benzene ring is labeled with 14C, where will the label be in the product? A
-
What starting materials are required for the synthesis of the following compounds, using the Fischer indole synthesis? (Hint: See Problem 50.) a. b. c. CH2CH3 CH2CH3
-
a. Which is a stronger base, CH3COO- or HCOO-? (The pKa of CH3COOH is 4.8; the pKa of HCOOH is 3.8.) b. Which is a stronger base, HO- or NH2? (The pKa of H2O is 15.7; the pKa of NH3 is 36.) c. Which...
-
James Halperin, co-chair of Heritage Auctions, was interviewed about his experiences auctioning rare coins. Noting that when the auction house first opened in the 1970s, I used to proofread every...
-
An editorial on Bloomberg. com criticized President Bidens Buy American policy. The editorial argued that the . . . problem is that protectionism sets the US at odds with its international...
-
An article in the Wall Street Journal explained that despite rising national security tensions between the United States and China in 2023, trade between the two nations was rising. China is a...

Study smarter with the SolutionInn App


