Consider the H, molecule with atoms HA and Hg separated by a distance R: HA-Ha. The...
Fantastic news! We've Found the answer you've been seeking!
Question:
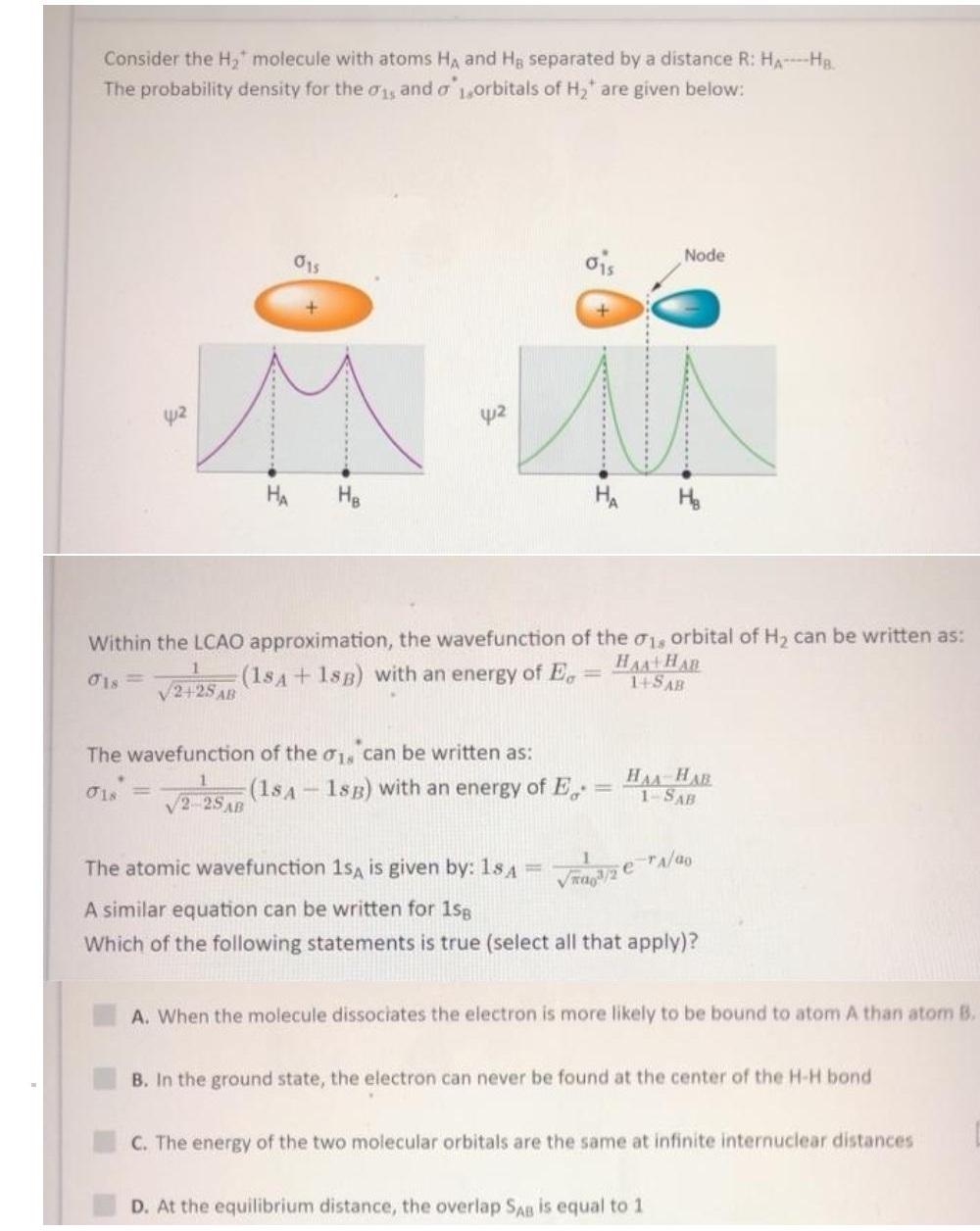
Transcribed Image Text:
Consider the H, molecule with atoms HA and Hg separated by a distance R: HA-Ha. The probability density for the 0y and o 1,orbitals of H, are given below: ois Node 015 42 42 HA HA H. Within the LCAO approximation, the wavefunction of the 01, orbital of H, can be written as: (18A+ 1sB) with an energy of E, HAA+HAR 14SAB V2+25 AB The wavefunction of the o1, can be written as: (1sA - 1sB) with an energy of E, HAA HAR 1-SAB O1s = V2-25AB The atomic wavefunction 1sA is given by: 1s A %3D Vra/2 erA/ao A similar equation can be written for 1sg Which of the following statements is true (select all that apply)? A. When the molecule dissociates the electron is more likely to be bound to atom A than atom B. B. In the ground state, the electron can never be found at the center of the H-H bond C. The energy of the two molecular orbitals are the same at infinite internuclear distances D. At the equilibrium distance, the overlap SAB is equal to 1 ....... Consider the H, molecule with atoms HA and Hg separated by a distance R: HA-Ha. The probability density for the 0y and o 1,orbitals of H, are given below: ois Node 015 42 42 HA HA H. Within the LCAO approximation, the wavefunction of the 01, orbital of H, can be written as: (18A+ 1sB) with an energy of E, HAA+HAR 14SAB V2+25 AB The wavefunction of the o1, can be written as: (1sA - 1sB) with an energy of E, HAA HAR 1-SAB O1s = V2-25AB The atomic wavefunction 1sA is given by: 1s A %3D Vra/2 erA/ao A similar equation can be written for 1sg Which of the following statements is true (select all that apply)? A. When the molecule dissociates the electron is more likely to be bound to atom A than atom B. B. In the ground state, the electron can never be found at the center of the H-H bond C. The energy of the two molecular orbitals are the same at infinite internuclear distances D. At the equilibrium distance, the overlap SAB is equal to 1 .......
Expert Answer:
Answer rating: 100% (QA)
Statement A FALSE Because the probability density function is symm... View the full answer

Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these chemistry questions
-
which of the following statements is true select one a. an employer cannot be held liable for a tort that an employee deliberately committed b. vicarious liability supports tort law's compensation...
-
Consider two protons (charge +e), separated by a distance of 2.0 1015 m (as in a typical atomic nucleus). The electric force between these protons is equal in magnitude to the gravitational force on...
-
The air between two parallel plates separated by a distance d = 20 mm is ionized by X-ray radiation. Each plate has an area S = 500 cm2. Find the concentration of positive ions if at a voltage V =...
-
Comparative financial statements of the Boeckman Company for 2009 and 2010 are as follows: Comparative Balance Sheets Comparative Income Statements Additional information: The Boeckman Company is...
-
Express the function in the form f o g oh. a. R(x) = x - 1 b. H(x) = sec4 (x)
-
Determine the position of the center of mass of the baton shown in Figure P6.41, taking the origin of your coordinate axis to be \((a)\) the center of the larger ball, \((b)\) the center of the...
-
The rigid bar is supported by the pin-connected rod CB that has a cross-sectional area of 500 mm2 and is made of A-36 steel. Determine the vertical displacement of the bar at B when the load is...
-
Brooklyn Cabinets is a manufacturer of kitchen cabinets. The two cabinetry styles manufactured by Brooklyn are contemporary and farmhouse. Contemporary style cabinets sell for $90 and farmhouse style...
-
if 2 ^ a = 5 and 2 ^ b = 3 , what is the value of log 3 ( 1 0 )
-
A manager of a store that sells and installs spas wants to prepare a forecast for January, February, and March of next year. Her forecasts are a combination of trend and seasonality. She uses the...
-
You want to sell your car for SAR 75,000/-. You assess the probability of getting an offer on your car of SAR 65,000 (or higher) to be 60%, based on recent car sales in your area. Suppose that you...
-
Using a company of your choice, evaluate their current service using the three Ps (people, process and physical evidence). Make recommendations as to how to develop these elements to enhance service...
-
The income statement for California Communications follows. Assume California Communications signed a three-month, 9%, $3,000 note on June 1, 2024, and that this was the only note payable for the...
-
What might be the most useful segmentation bases for the mobile phone market?
-
How do family members influence each others buying behaviour?
-
Under what circumstances would qualitative research be more appropriate than quantitative research?
-
1. A company (the producer) supplies microprocessors to a manufacturer(the consumer) of electronic equipment. The microprocessors are supplied in batches of 50. The consumer regards a batch as...
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
Yosemite Falls in California is about 740 m high. (a) What average power would it take for a 70-kg person to hike up to the top of Yosemite Falls in 1.5 h? (b) The human body is about 25% efficient...
-
A star is moving away from Earth at a speed of 2.4 108 m/s. Light of wavelength 480 nm is emitted by the star. What is the wavelength as measured by an Earth observer?
-
A strip of copper 2.0 cm wide carries a current I = 30.0 A to the right. The strip is in a magnetic field B = 5.0 T into the page. (a) What is the direction of the average magnetic force on the...
-
Which of the following is a defined benefit plan? A. 403(b). B. 412(i). C. 457(b). D. 457(f).
-
An SEP has which characteristic? A. Loans and hardship withdrawals are available. B. Age-weighting or cross-testing is permitted. C. Social Security integration is permitted. D. Employer matching is...
-
Which type of funding vehicle is not approved for 403(b) plans? A. Mutual fund. B. Variable annuity contract. C. Individual securities. D. Fixed annuity contract.

Study smarter with the SolutionInn App


