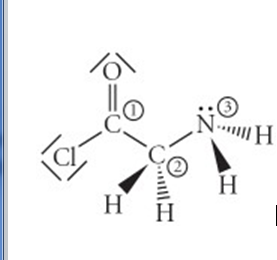
Consider the molecule below. Determine the hybridization at each of the 3 labeled atoms. Along with the
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider the molecule below. Determine the hybridization at each of the 3 labeled atoms.
Along with the hybridization determine how many σ bonds and how many π bonds each of those 3 atoms can have.
I'm really not sure how to do this problem? I don't get how to tell how many σ bonds and π bonds there are? I also don't understand how to get the hybridization?
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Posted Date:




