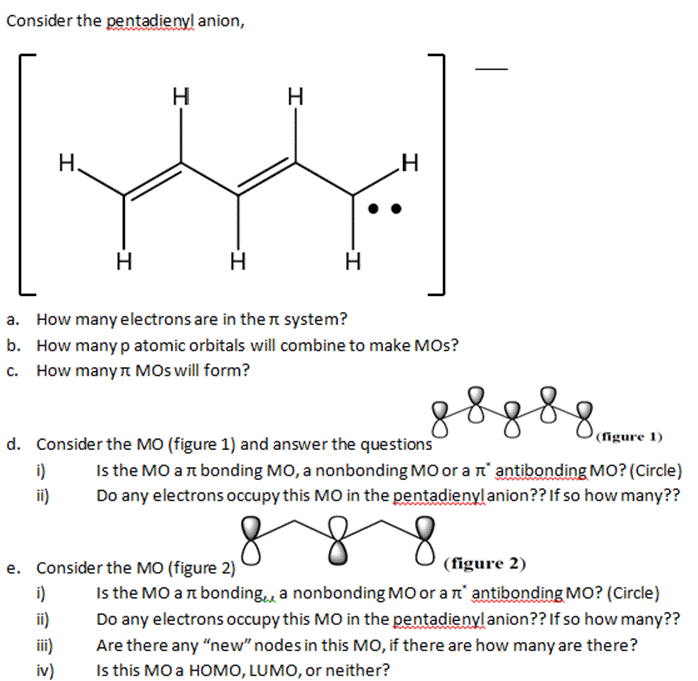
Consider the pentadieny! anion, i) I ii) H. -I i) ii) iii) iv) H H H...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the pentadieny! anion, i) I ii) H. -I i) ii) iii) iv) H H H a. How many electrons are in the system? b. How many p atomic orbitals will combine to make MOS? c. How manyл MOs will form? H H d. Consider the MO (figure 1) and answer the questions H e. Consider the MO (figure 2) g 8 g s g m (figure 1) Is the MO art bonding MO, a nonbonding MO or a π antibonding MO? (Circle) Do any electrons occupy this MO in the pentadienylanion?? If so how many?? (figure 2) Is the MO ar bonding a nonbonding MO or a π antibonding MO? (Circle) Do any electrons occupy this MO in the pentadienyl anion?? If so how many?? Are there any "new" nodes in this MO, if there are how many are there? Is this MO a HOMO, LUMO, or neither? Consider the pentadieny! anion, i) I ii) H. -I i) ii) iii) iv) H H H a. How many electrons are in the system? b. How many p atomic orbitals will combine to make MOS? c. How manyл MOs will form? H H d. Consider the MO (figure 1) and answer the questions H e. Consider the MO (figure 2) g 8 g s g m (figure 1) Is the MO art bonding MO, a nonbonding MO or a π antibonding MO? (Circle) Do any electrons occupy this MO in the pentadienylanion?? If so how many?? (figure 2) Is the MO ar bonding a nonbonding MO or a π antibonding MO? (Circle) Do any electrons occupy this MO in the pentadienyl anion?? If so how many?? Are there any "new" nodes in this MO, if there are how many are there? Is this MO a HOMO, LUMO, or neither?
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
How many electrons are in the valence d orbitals in these transition-metal ions? (a) Co3+, (b) Cu+, (c) Cd2+, (d) Os3+,.
-
(a) How many electrons are in one coulomb? (b) How many coulombs are in one mole of charge?
-
How many electrons are shared in a double covalent bond?
-
1. If net profit is $47,025; depreciation is $2,200; accounts receivable increases $5,056; accounts payable increases $4,673; and inventory declines $850; what is the operating cash flow for the...
-
An insurance company is updating its payouts and cost structure for their insurance policies. Of particular interest to them is the risk analysis for customers currently on heart or blood pressure...
-
Larry Backwater, a resident primary producer, had the following receipts and payments during the 2019/20 year: RECEIPTS Sale of Sheep (13,400 head) - note 2 Wool Sales note 3 Capital Insurance...
-
How would a company know that its product or service was preferred in the marketplace? What could a competitor do to erode this favorable position?
-
Wooden boxes are commonly used for the packaging and transportation of mangoes. A convenience store in Morganville, New Jersey, regularly buys mangoes from a wholesale dealer. For every shipment, the...
-
Suppose Union University has spent $10,000 on an option to purchase land for new dormitories at a price of $100,000. Also assume that it is later offered an equally attractive site for $95,000. What...
-
The airline industry defines an on time flight as one that arrives within 15 minutes of its scheduled time. The following table shows the number of on time and late flights leaving Philadelphia and...
-
The number of models considered when using best subset selection is (where p is the number of candidate predictors): p models, since there are p covariates 2^p p(p+1) 2 p(p+1) 2 +1
-
A bank loan granted considers the interest rate of 48% annually, compounded quarterly. Calculate the final value to be covered if the amount borrowed is $71,000 over 4 years.?
-
A solid - gold jewerly box was found in a the underwater palace of Cleopatra just off the shore of Alexandria. The length of the rectangular box is 3 5 centimeters less than the triple the width. The...
-
Find the 4th term of the sequences= =(n-2) (3)"-1 Blank 1
-
If you were told that a Multiple R from a regression analysis was .632 and the R Square is .16, what would that tell you about the data? Please be as specific and details as possible.
-
The weight (in pounds) of a certain population of men is normally distributed. A random sample of 16 men taken from the population yields a sample mean weight of 168 with a sample variance of 225....
-
11. Create an uninitialized data declaration for an 8-bit unsigned integer? 12. Which data type can hold a 32-bit signed integer? 13. Declare an unsigned 16-bit integer variable named wArray that...
-
Medi-Exam Health Services, Inc. (MEHS), located in a major metropolitan area, provides annual physical screening examinations, including a routine physical, EKG, and blood and urine tests. MEUS's...
-
A 53.8-mg sample of sodium perchlorate contains radioactive chlorine-36 (whose atomic mass is 36.0 amu). If 29.6% of the chlorine atoms in the sample are chlorine-36 and the remainder are naturally...
-
The crude copper that is subjected to electrorefining contains tellurium as an impurity. The standard reduction potential between tellurium and its lowest common oxidation state, Te4+, is Te4+(aq) +...
-
Butylated hydroxytoluene (BHT) has the following molecular structure: It is widely used as a preservative in a variety of foods, including dried cereals. Based on its structure, would you expect BHT...
-
Write a numerical algorithm to solve Eq. 8.71 for \(A_{n}\). Obtain lift coefficients for \(h / 2 b=2\) and \(\mathrm{AR}=6,10\) at \(\mathrm{AoA}=12\). Eq. 8.71 Asin no 1 (y) 1 ban b 4h2b2 (cos...
-
Using (8.76) find the chord wise variation of the lift as \(L^{\prime}(x)\). Eq 8.76 1 za 2hg ax ax ax - (x)- aza
-
Why are none of the bulbs in Figure 31. 24 lit? Data from Figure 31. 24 Figure 31.24 (1) (iii) NNN

Study smarter with the SolutionInn App


