Consider the stirred-tank reactor shown in Fig. P5-5. The reaction occurring is A ? B and it
Fantastic news! We've Found the answer you've been seeking!
Question:
Consider the stirred-tank reactor shown in Fig. P5-5.
The reaction occurring is
A ? B
and it proceeds at a rate
r = kCo
where r = (moles A reacting)/(volume)(time)
k = reaction rate constant
Co (t) = concentration of A in reactor at any time t (mol A/volume)
V = volume of mixture in reactor
Further, let
F = constant feed rate, volume/time
Ci (t) = concentration of A in feed stream, moles/volume
Assuming constant density and constant volume V, derive the transfer function relating the concentration in the reactor to the feed-stream concentration. Prepare a block diagram for the reactor. Sketch the response of the reactor to a unit-step change in Ci. 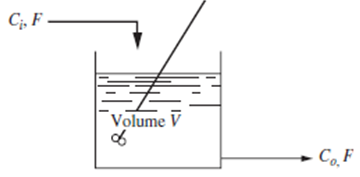
Related Book For 

Probability and Random Processes With Applications to Signal Processing and Communications
ISBN: 978-0123869814
2nd edition
Authors: Scott Miller, Donald Childers
Posted Date:




