Transitions between energy levels in atoms or molecules depend upon the satisfaction of selection rules. The...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
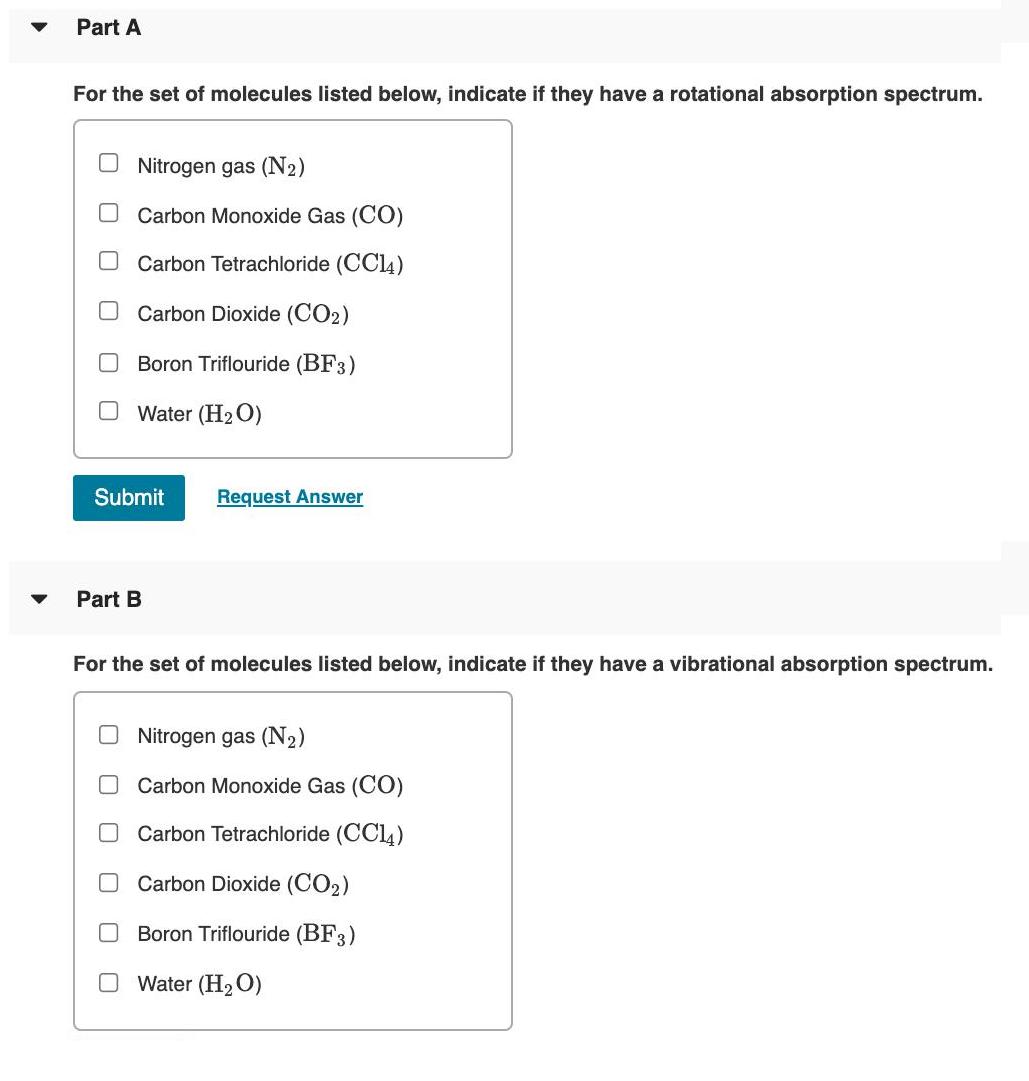
Transitions between energy levels in atoms or molecules depend upon the satisfaction of selection rules. The strength for a transition from an initial state with wavefunction b; to a final state with wavefunction f is given by the transition moment integral: Ix where u, is the transition moment operator. For rotational and vibrational spectroscopy the dipole moment plays a critical role in this operator. The dipole moment can be expanded about the value at the equilibrium internuclear distance: 20 dQ+... Qe where dQ is the change in the geometry of the molecule. For a molecule to have a rotational absorption spectrum the permanent dipole moment must be non-zero and for a molecule to have a vibrational absorption spectrum the change in dipole moment with changing geometry must be non-zero. Vibrational modes are Raman allowed if there is a change in polarizability u = le + with changing molecular geometry: (옮) 40 da a = ao+ aQ Because this leads to an induced dipole in the molecule: %3D Min = aE Part A For the set of molecules listed below, indicate if they have a rotational absorption spectrum. O Nitrogen gas (N2) Carbon Monoxide Gas (CO) Carbon Tetrachloride (CC14) OCarbon Dioxide (CO2) Boron Triflouride (BF3) O Water (H2O) Submit Request Answer Part B For the set of molecules listed below, indicate if they have a vibrational absorption spectrum. O Nitrogen gas (N2) Carbon Monoxide Gas (CO) Carbon Tetrachloride (CCl4) Carbon Dioxide (CO2) Boron Triflouride (BF3) O Water (H2O) O O O O Transitions between energy levels in atoms or molecules depend upon the satisfaction of selection rules. The strength for a transition from an initial state with wavefunction b; to a final state with wavefunction f is given by the transition moment integral: Ix where u, is the transition moment operator. For rotational and vibrational spectroscopy the dipole moment plays a critical role in this operator. The dipole moment can be expanded about the value at the equilibrium internuclear distance: 20 dQ+... Qe where dQ is the change in the geometry of the molecule. For a molecule to have a rotational absorption spectrum the permanent dipole moment must be non-zero and for a molecule to have a vibrational absorption spectrum the change in dipole moment with changing geometry must be non-zero. Vibrational modes are Raman allowed if there is a change in polarizability u = le + with changing molecular geometry: (옮) 40 da a = ao+ aQ Because this leads to an induced dipole in the molecule: %3D Min = aE Part A For the set of molecules listed below, indicate if they have a rotational absorption spectrum. O Nitrogen gas (N2) Carbon Monoxide Gas (CO) Carbon Tetrachloride (CC14) OCarbon Dioxide (CO2) Boron Triflouride (BF3) O Water (H2O) Submit Request Answer Part B For the set of molecules listed below, indicate if they have a vibrational absorption spectrum. O Nitrogen gas (N2) Carbon Monoxide Gas (CO) Carbon Tetrachloride (CCl4) Carbon Dioxide (CO2) Boron Triflouride (BF3) O Water (H2O) O O O O
Expert Answer:

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these chemistry questions
-
A sample of gas undergoes a transition from an initial state a to a final state b by three different paths (processes), as shown in the p-V diagram in Figure where Vb = 5.00Vi. The energy transferred...
-
Transitions between energy levels in atoms or molecules depend upon the satisfaction of selection rules. The strength for a transition from an initial state with wavefunction b; to a final state with...
-
Suppose stock returns can be explained by a two-factor model. The firm-specific risks for all stocks are independent. The following table shows the information for two diversified portfolios: 1 2...
-
In Exercises find the given higher-order derivative. 2 "(x) = 2 / f(x) X
-
Construct a spreadsheet for which you can input up to five strike prices and quantities of put and call options bought or sold at those strikes, and which will automatically construct the total...
-
What criteria might managers use to guide cost-allocation decisions? Which are the dominant criteria?
-
What is the purpose of practice aids in forensic and valuation services?
-
For 2014, Lindell Company reported its most significant decline in net income in years. At the end of the year, H. Finn, the president, is presented with the following condensed comparative income...
-
A combined solar and auxiliary energy system is used to meet the same load as in Example 12.5. The total cost of the system to cover 65% of the load (solar fraction) is $20,000. The owner will pay a...
-
Jessie Howe is examining Met Companys accounts receivable balance and has decided to use mean- per- unit estimation. Mets accounts receivable were recorded at $ 650,000 and comprised 2,000 individual...
-
2) Considers pros and cons to deal with non-sustainable debt [5 pts]: a. Government asks the central bank to buy government bonds as they come to maturity b. Default c. Increase in taxes and/or...
-
Discuss the pros and cons of diversification as a rationale for mergers.
-
Why should investors be concerned about a firms outstanding warrants and convertibles?
-
Explain why it is imperative to measure the correct construct? What could go wrong if the correct constructs are not used in a research project?
-
Give two examples of how tax considerations can motivate mergers.
-
Define synergy. Is synergy a valid rationale for mergers? Describe several situations that might produce synergistic gains.
-
(9) To determine what/how a community determines levels of public goods, we discussed the application of three theories (or principles) and how they interact with each other. Explain these three and...
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
An air-standard Diesel cycle absorbs 1.500 J mol-1 of heat OL Volume
-
Large quantities of liquefied natural gas (LNG) are shipped by ocean tanker. At the unloading port provision is made for vaporization of the LNG so that it may be delivered to pipelines as gas. The...
-
The following reaction reaches equilibrium at 650C and atmospheric pressure: C6H5CH : CH2 (g) + H2 (g) C6H5 . C2H5 (g) If the system initially contains 1.5 mol H2 for each mole of styrene what is...
-
We can make a static measurement to deduce the spring constant to use in the model. If a \(61 \mathrm{~kg}\) woman stands on a low wall with her full weight on the ball of one foot and the heel free...
-
A \(1.00 \mathrm{~kg}\) block is attached to a horizontal spring with spring INT constant \(2500 \mathrm{~N} / \mathrm{m}\). The block is at rest on a frictionless surface. A \(10.0 \mathrm{~g}\)...
-
If, during a stride, the stretch causes her center of mass to lower by \(10 \mathrm{~mm}\), what is the stored energy? A. \(3.0 \mathrm{~J}\) B. \(6.0 \mathrm{~J}\) C. \(9.0 \mathrm{~J}\) D. \(12...

Study smarter with the SolutionInn App


