Constants Periodic Table Calculate the energy of a photon of electromagnetic radiation at each of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
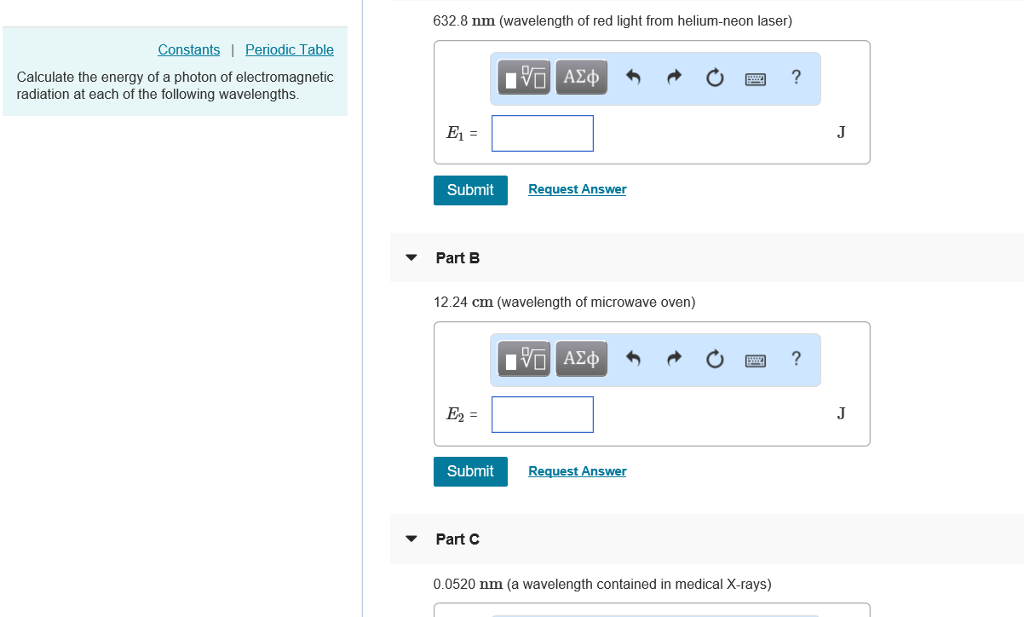
Transcribed Image Text:
Constants Periodic Table Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 632.8 nm (wavelength of red light from helium-neon laser) E₁ = Submit Part B E₂ = 12.24 cm (wavelength of microwave oven) Submit 15. ΑΣΦ Part C Request Answer —| ΑΣΦ Request Answer 0.0520 nm (a wavelength contained in medical X-rays) ? ? J J Constants Periodic Table Calculate the energy of a photon of electromagnetic radiation at each of the following wavelengths. 632.8 nm (wavelength of red light from helium-neon laser) E₁ = Submit Part B E₂ = 12.24 cm (wavelength of microwave oven) Submit 15. ΑΣΦ Part C Request Answer —| ΑΣΦ Request Answer 0.0520 nm (a wavelength contained in medical X-rays) ? ? J J
Expert Answer:
Answer rating: 100% (QA)
Energy of Photon is given by E ho E hxc Paszt A E E Part B O 0 hc A632810 m C ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
(a) Calculate the energy of a photon of electromagnetic radiation whose frequency is 6.75 1012s-1. (b) Calculate the energy of a photon of radiation whose wavelength is 322 nm. (c) What wavelength...
-
(a) Calculate the energy of a photon of electromagnetic radiation whose frequency is 2.94 10 14 s-1. (b) Calculate the energy of a photon of radiation whose wavelength is 413 nm. (c) What wavelength...
-
1. Find the energy of a photon of electromagnetic radiation with frequency 9.20 x 1016 Hz. 2. Find the energy of a red photon. 3. Find the energy of a blue photon. 4. Find the energy of a yellow...
-
On September 30, 2021, Antagonia Real Estate Limited's general ledger showed the following balances: Accounts payable Accounts receivable Advertising expenses Cash Common shares Income tax expense...
-
Grate Care Company specializes in producing products for personal grooming. The company operates six divisions, including the Hair Products Division. Each division is treated as an investment center....
-
How can exchange rates change to reduce wage differences between countries?
-
Would you rather receive \($1,000\) today or \($2,000\) in 10 years if the discount rate is 8 percent?
-
Decorative Steel, Inc., began August with 55 units of iron inventory that cost $35 each. During August, the company completed the following inventory transactions: Requirements 1. Prepare a perpetual...
-
Direct materials purchases budget Tobin's Frozen Pizza Inc. has determined from its production budget the following estimated production volumes for 12" and 16" frozen pizzas for November: Budgeted...
-
Recording a Business Combination Petra Corporation paid $ 8 million in cash to acquire the assets and liabilities of Salisbury Company. Petra also agreed to make an additional cash payment in the...
-
Write a C++ program to insert or delete characters in a buffer. You need to develop an abstract data type for a buffer in the text editor program that implements the following operations using linked...
-
Commercial airliners normally cruise at relatively high altitudes \((30,000\) to \(35,000 \mathrm{ft}\) ). Discuss how flying at this high altitude (rather than 10,000 ft, for example) can save fuel...
-
At some point for air flow in a duct, \(p=20 \mathrm{psia}, T=500^{\circ} \mathrm{R}\), and \(V=500 \mathrm{ft} / \mathrm{s}\). Can a normal shock occur at this point?
-
The landing speed of a winged aircraft such as the Space Shuttle is dependent on the air density. By what percent must the landing speed be increased on a day when the temperature is \(110^{\circ}...
-
Calculate the speed of sound in air, helium, and hydrogen. The temperature is \(70^{\circ} \mathrm{F}\).
-
A trout jumps, producing waves on the surface of a 0.8 -m-deep mountain stream. If it is observed that the waves do not travel upstream, what is the minimum velocity of the current?
-
Critically analyze the superiority of the demand-based model adopted by Social Ventures Australia over a supply-based model.
-
Using the theoretical sampling strategy, how many samples of size 4 (n = 4) can be drawn from a population of size: (a) N = 5? (b) N = 8? (c) N = 16? (d) N = 50?
-
Is there any correlation between the anions that form when each of the strong acids in Table 4.2 dissociates and the anions that normally form soluble ionic compounds (Table 4.1)? Which anions if any...
-
(a) Which of these three lines represents the effect of concentration on the percent ionization of a weak acid? (b) Explain in qualitative terms why the curve you chose has the shape it does. ZI Acid...
-
Explain how the existence of line spectra is consistent with Bohrs theory of quantized energies for the electron in the hydrogen atom.
-
The following trial balance was extracted from the books of A Scholes at the close of business on 28 February 19X7. Notes: (a) Stock 28 February 19X7 3,510. (b) Wages and salaries accrued at 28...
-
Express the vector \(\mathbf{v}=(1,2,3)\) as a linear combination of the vectors \(\mathbf{a}_{1}=(1,1,1), \mathbf{a}_{2}=(1,0,-2)\), and \(\mathbf{a}_{3}=(2,1,0)\).
-
T Morgan, a sole trader, extracted the following trial balance from his books at the close of business on 31 March 19X9: Notes: a) Stock 31 March 19X9 4,290. b) Wages and salaries accrued at 31 March...

Study smarter with the SolutionInn App


