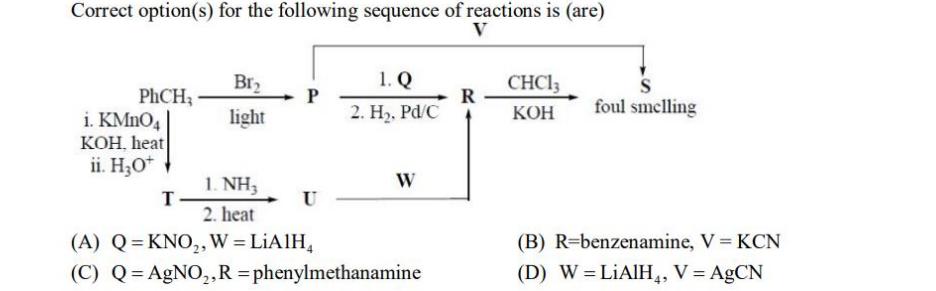
Correct option(s) for the following sequence of reactions is (are) V PhCH, i. KMnO4 KOH. heat...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Correct option(s) for the following sequence of reactions is (are) V PhCH, i. KMnO4 KOH. heat ii. H₂O+ Br₂ light T P 1. NH3 2. heat (A) Q=KNO₂, W = LiAlH₂ (C) Q=AgNO₂, R =phenylmethanamine 1. Q 2. H₂, Pd/C U W R CHC13 KOH foul smelling (B) R-benzenamine, V KCN (D) W = LiAlH4, V = AgCN Correct option(s) for the following sequence of reactions is (are) V PhCH, i. KMnO4 KOH. heat ii. H₂O+ Br₂ light T P 1. NH3 2. heat (A) Q=KNO₂, W = LiAlH₂ (C) Q=AgNO₂, R =phenylmethanamine 1. Q 2. H₂, Pd/C U W R CHC13 KOH foul smelling (B) R-benzenamine, V KCN (D) W = LiAlH4, V = AgCN
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The following sequence of virtual page numbers is encountered in the course of execution on a computer with virtual memory: 3 4 2 6 4 7 1 3 2 6 3 5 1 2 3 Assume that a least recently used page...
-
The herbicide trifluralin is prepared by the following sequence of reactions. Identify compound A and deduce the structure of trifluralin. CF3 (CH,CH,CHNH Trifluralin HCompound A CH CIFN204) Cl
-
A segment of human DNA has the following sequence of nucleotide pairs: When this segment of DNA is transcribed by RNA polymerase, what will be the sequence of nucleotides in the RNA transcript?...
-
Evaluate the geometric series or state that it diverges. 8 00 k=0 2k 75
-
Does assumption of the risk apply to jet skiing? On a warm California day, there were about 30 personal watercraft (jet skis) operating on Coyote Lake. The weather was fair and visibility good....
-
Show that the group velocity can be written as n + w(dn/dw)
-
What is the implication of the statement "the books are not in balance"? What corrective action should be taken?
-
Use the information in the adjusted trial balance reported in Exercise 4-4 to compute the current ratio as of the balance sheet date (round the ratio to two decimals). Interpret the current ratio for...
-
Circle T is shown below. M T N P Nina measured the length of line segment MP as 20 centimeters (cm). What is the length of line segment NT?
-
Two flow control loops axe shown in the drawing. Indicate whether each system is either a feedback or a feed-forward control system. Justify your answer. It can be assumed that the distance between...
-
A manufacturing plant is producing the following products using available raw materials shown in the below Table. Resource usage per unit product Resource Available resource Product 1 Product 2 Raw...
-
Using the Ethical Systems Model Survey , complete an assessment of Uber's internal reporting system. What do you see as strengths and/or weaknesses of Uber's process?
-
Why is performance management necessary? 2) What can HR do to help make this process effective for the organization? 3) Discuss what benefits are most important to American employees today.
-
Make your own shared poem. Describe your process to coming up with it?
-
INSTRUCTIONS research one aspect/topic of occupational health and safety law. Once completed, make an essay. The written report: Your report should be within three to ten pages. Your report should...
-
How does Amazon work within the following management function: Organizing How is work organized, delegated, and managed? Describe role of human resource management at Amazon.
-
Richard and Pauline Jackson have a small mowing and gardening business in which they have been working for the last eight years. As it is only the two of them in the business they operate as a...
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
Show how the following compounds could be prepared, using only the indicated starting material as the source of carbon: a. b. c. d. CH CH3CCH3 from CH3CHCH3 CH3 CH3CH-CCH3 O CH3 CH3C CHCH3 CH CH2CH
-
Which reactant in each of the following pairs will undergo an elimination reaction more rapidly? Explain your choice. a. b. - - (CH3)C 20 CH, CH30- CH3OH CH3 Br 3 CH3o Br CH3OH
-
Which monomer and which type initiator would you use to synthesize each of the following polymers? a. b. c. ¬CH2CH2OCH2CH2O¬ d. CHs CH3 CHs CH3 CH3 CH3 CH,CH-CH-CH- CH CHCH CH COCH COCH
-
Trans Clothing Alterations began operations on 1 August 2024 and completed the following transactions during the first month. 1. Tran deposited \($18\) 000 of her personal funds in a current account...
-
Finesse Fitness was established on 1 April 2024 with an initial investment of $60000 by the owner, Daniel Hewitt. During the first few months of business, the owner employed a student studying...
-
Jason Vu offers tutoring services to first-year university students. He has set up a sole proprietorship business named JV Tutoring. Jason has collected the following information relating to his...

Study smarter with the SolutionInn App



