Question: Create a stream table showing for each stream the mass flow rates of all components, the temperature and the pressure using the attached chart. Project
Create a stream table showing for each stream the mass flow rates of all components, the temperature and the pressure using the attached chart.


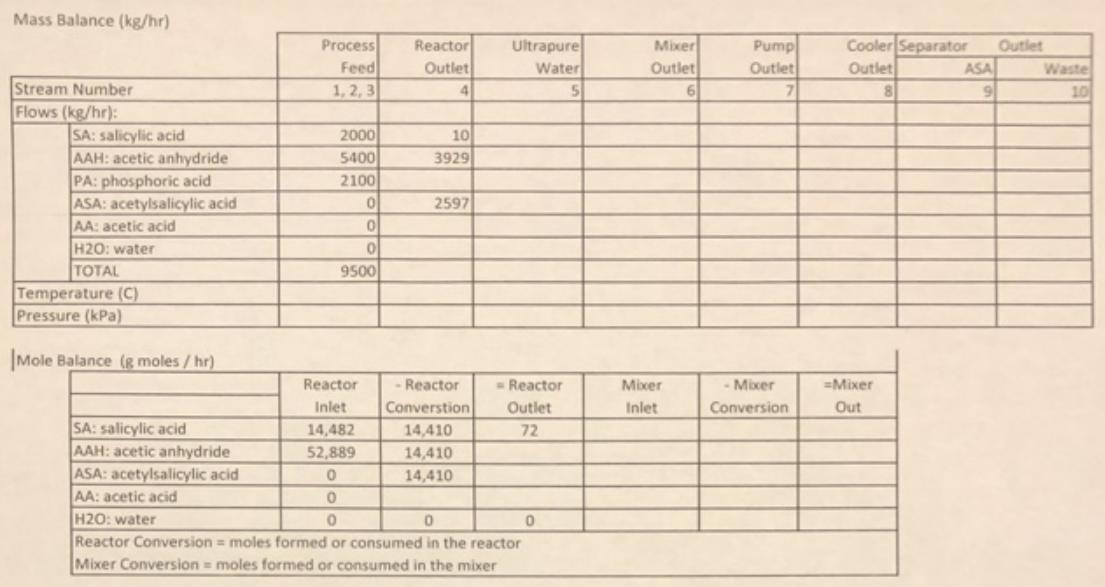
Project Description In the manufacture of aspirin, acetylsalieylic acid (ASA, the active ingredient in aspirin) is synthesized using the reaction salicylic acid + acetic anhydride acetylsaliylic acid + acetic acid The presence of phosphoric acid catalyzes the irreversible reaction, which proceeds with kinetics that are first-order with respect to salicylic acid concentration (koetin0.5 sl), Further, the reaction is exothermic (AHeact-85,800 J/gmol of salicylic acid reacted). A simplified process is described below: The following three streams will be continuously fed to a reactor at 70C: Flow rate 2000 kg/hr 5000 L/hr 1250 L/hr Feed Stream Component Crystalline salicylic acid Liquid acetic anhydride (pure) Liquid concentrated (95%) phosphoric acid The salicylic acid will enter the reactor as a crystalline solid but will dissolve in the acetic anhydride. The reactor is a continuously stirred tank reactor, where 99.5% of the salicylic acid will be converted to acetylsalicylic acid (also dissolved). Furthermore, the contents of the reactor will be maintained at 70C in the reactor by a cooling jacket around the reactor (carrying cooling water as needed). The outlet stream from the reactor will flow to a mixer where it will be joined by an additional stream of ultrapure water (22,000 L/hr, 25C). The water will react with the excess acetic anhydride to form more acetic acid, according to the reaction acetic anhydride + water + 2(acetic acid) This reaction proceeds to 100% conversion in the mixer, so the sizing of a "reactor" for this step is not necessary. Further, this reaction is also exothermic, but the extent of this reaction is small enough in this case to justify neglecting the heat of the reaction. The mixed product stream leaving the mixer will enter a centrifugal pump, which will boost the pressure to the value needed to pass through the remainder of the process (leaving the final filtration sequence at atmospheric pressure). The stream leaving the pump will enter a product cooler (a heat exchanger) in which it will be cooled to 25C using cooling water as the other stream in the exchanger. The water addition and cooling step will promote the crystallization of the acetylsalicylic acid (and any unreacted salicylic acid), and the stream will then enter a filtration sequence to separate the crystals (aspirin stream) from the other byproducts (acetic acid byproduct stream, which is sold).
Step by Step Solution
3.44 Rating (144 Votes )
There are 3 Steps involved in it
To create a stream table for the process described well fill in details for each stream including ma... View full answer

Get step-by-step solutions from verified subject matter experts


