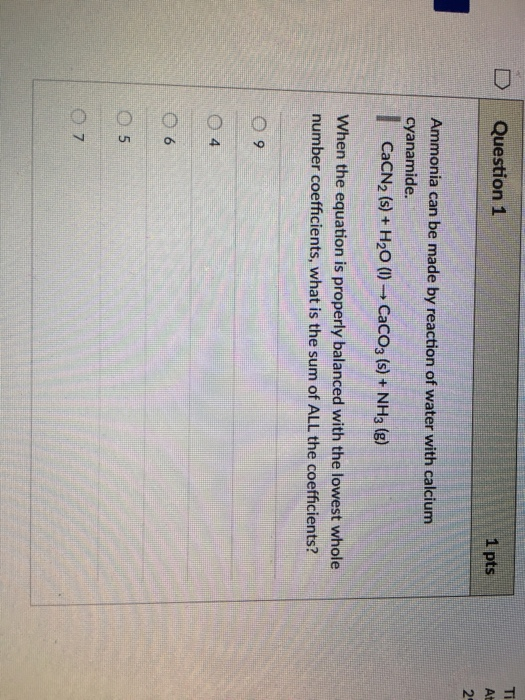
D Question 1 Ammonia can be made by reaction of water with calcium cyanamide. CaCN (s)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
D Question 1 Ammonia can be made by reaction of water with calcium cyanamide. CaCN₂ (s) + H₂O (1)→ CaCO3 (s) + NH3 (g) When the equation is properly balanced with the lowest whole number coefficients, what is the sum of ALL the coefficients? 9 A 06 5 1 pts 07 Ti At D Question 1 Ammonia can be made by reaction of water with calcium cyanamide. CaCN₂ (s) + H₂O (1)→ CaCO3 (s) + NH3 (g) When the equation is properly balanced with the lowest whole number coefficients, what is the sum of ALL the coefficients? 9 A 06 5 1 pts 07 Ti At
Expert Answer:
Answer rating: 100% (QA)
Answer is 7 CaN S H0 1 Ca03 3 NH 3 NH3 7 balance chemical different atoms should ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
A steel part can be made by powder metallurgy (compacting iron powder particles and sintering to produce a solid) or by machining from a solid steel block. Which part is expected to have the higher...
-
The explosive TNT (2,4,6-trinitrotoluene) can be made by nitrating toluene with a mixture of nitric and sulfuric acids, but the reaction conditions must gradually be made more severe as the nitration...
-
The fertilizer ammonium sulfate can be made by using sulfuric acid to neutralize a solution of ammonia in water. Give the overall equation of the process.
-
Prepare journal entries to record the following grant-related transactions of an enterprise fund activity. Explain how these transactions should be reported in the enterprise funds financial...
-
Kramer, CPA, consulted an independent appraiser regarding the valuation of fine art for a not- for- profit museum. Consultation with the appraiser in this case would a. Be considered as exercising...
-
A wheel has a thin 3.0-kg rim and four spokes each of mass 1.2 kg. Find the kinetic energy of the wheel when it rolls at 6 m/s on a horizontal surface.
-
Nationwide Insurance developed a program to recruit new insurance agents by offering planning, training, and start-up financing to build self-sustaining agencies. These new agents would be...
-
Helix Company produces costumes used in the television and movie industries. Recently the company received an ongoing order for Samurai robes to be worn in an upcoming Japanese historical action...
-
Define the relational model. What is a relational database management system (DBMS)?
-
Blossom Corporation is preparing the comparative financial statements to be included in the annual report to stockholders. Blossom employs a fiscal year ending May 31. Income from operations before...
-
Visit the link given below See IT sourcing and cloud strategy Find 2 peer-reviewed articles from academic journals on information technology sourcing. Summarize each article and compare and contrast...
-
2. Convert 9210 to Binary, Octal and Hexadecimal using repeated division. Show your work. Answer: Binary: Octal: Hexadecimal:
-
The purpose of this project is to demonstrate your grasp of the concepts you have learned throughout the course and your ability to analyze the financial standings and history of a company. You...
-
B. Given a universal set of name strings as: Ali, Alan, Alice, Barb, Bob, Ben, Christie, Cora, Don, Dora, Fred, arranged in alphabetic order. Give a bit string representation of the following sets:...
-
what is the equation to represent the total cost? Units Cost High Activity Level 11,000 $14,800 Low Activity Level 7,000 $11,300 Change 4,000 $3,500 Unit Variable cost 0.875 Fixed cost 5175
-
John and Kent share profits and losses of 60% and 40%, respectively. The tax basis of each partner's interest in the partnership as of December 31, Year 1, was as follows: John $24,000 Kent 18,000...
-
find the yield-to-maturity (YTM) of the following bond of IOU Corp. These are the IOU Corp bond characteristics: -20 years left until maturity -A par value of $1,000 which will be paid at maturity -A...
-
Determine two different Hamilton circuits in each of the following graphs. A B F G
-
The first 25 years of the twentieth century were momentous for the rapid pace of change in scientists' understanding of the nature of matter. (a) How did Rutherford's experiments on the scattering of...
-
(a) Based on the lattice energies of MgCl2 and SrCl2 given in Table 8.2, what is the range of values that you would expect for the lattice energy of CaCl2? (b) Using data from Appendix C, Figure 7.9,...
-
Predict the chemical formula of the ionic compound formed between the following pairs of elements: (a) Al and F, (b) K and S, (c) Y and O, (d) Mg and N.
-
Use the magnitudes (Richter scale) of the earthquakes listed in Data Set 16 in Appendix B. In 1989, the San Francisco Bay Area was struck with an earthquake that measured 7.0 on the Richter scale....
-
Based on data from Data Set 17 in Appendix B, the words spoken in a day by women have a mean of 16,215 words and a standard deviation of 7301 words. Consider the women with an unusual word count in a...
-
Measures of Location The values of P 50 , Q 2 , and the median are found for the net incomes reported on all individual 1040 tax forms filed last year. What do those values have in common?

Study smarter with the SolutionInn App


