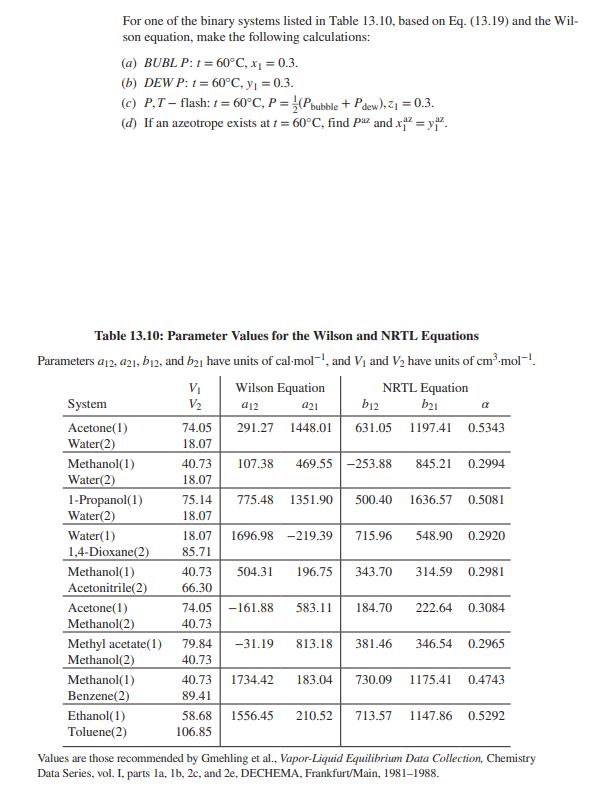
For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the Wil- son equation, make the following calculations: (a) BUBLP: 1 = 60°C, x₁ = 0.3. (b) DEW P: 1= 60°C, y₁ = 0.3. (c) P,T-flash: t=60°C, P = (Pbubble + Pdew), Z₁ = 0.3. (d) If an azeotrope exists at t=60°C, find Paz and x²=yiz. Table 13.10: Parameter Values for the Wilson and NRTL Equations Parameters a12, 421, b12, and b21 have units of cal-moll, and V₁ and V2 have units of cm³.mol-1. Wilson Equation System V₁ V2 a12 a21 b12 Acetone(1) 74.05 291.27 1448.01 631.05 NRTL Equation b21 1197.41 0.5343 α Water(2) 18.07 Methanol(1) 40.73 107.38 469.55 -253.88 845.21 0.2994 Water(2) 18.07 1-Propanol(1) Water(2) 75.14 775.48 1351.90 500.40 18.07 1636.57 0.5081 Water(1) 1,4-Dioxane(2) 18.07 1696.98 -219.39 85.71 715.96 548.90 0.2920 Methanol(1) Acetonitrile(2) 40.73 504.31 196.75 66.30 343.70 314.59 0.2981 Acetone(1) Methanol(2) 74.05 -161.88 583.11 184.70 40.73 222.64 0.3084 Methyl acetate(1) Methanol(2) 79.84 -31.19 813.18 40.73 381.46 346.54 0.2965 Methanol(1) Benzene(2) 40.73 1734.42 183.04 730.09 89.41 1175.41 0.4743 Ethanol(1) Toluene(2) 58.68 1556.45 210.52 106.85 713.57 1147.86 0.5292 Values are those recommended by Gmehling et al., Vapor-Liquid Equilibrium Data Collection, Chemistry Data Series, vol. I, parts la, lb, 2c, and 2e, DECHEMA, Frankfurt/Main, 1981-1988. For one of the binary systems listed in Table 13.10, based on Eq. (13.19) and the Wil- son equation, make the following calculations: (a) BUBLP: 1 = 60°C, x₁ = 0.3. (b) DEW P: 1= 60°C, y₁ = 0.3. (c) P,T-flash: t=60°C, P = (Pbubble + Pdew), Z₁ = 0.3. (d) If an azeotrope exists at t=60°C, find Paz and x²=yiz. Table 13.10: Parameter Values for the Wilson and NRTL Equations Parameters a12, 421, b12, and b21 have units of cal-moll, and V₁ and V2 have units of cm³.mol-1. Wilson Equation System V₁ V2 a12 a21 b12 Acetone(1) 74.05 291.27 1448.01 631.05 NRTL Equation b21 1197.41 0.5343 α Water(2) 18.07 Methanol(1) 40.73 107.38 469.55 -253.88 845.21 0.2994 Water(2) 18.07 1-Propanol(1) Water(2) 75.14 775.48 1351.90 500.40 18.07 1636.57 0.5081 Water(1) 1,4-Dioxane(2) 18.07 1696.98 -219.39 85.71 715.96 548.90 0.2920 Methanol(1) Acetonitrile(2) 40.73 504.31 196.75 66.30 343.70 314.59 0.2981 Acetone(1) Methanol(2) 74.05 -161.88 583.11 184.70 40.73 222.64 0.3084 Methyl acetate(1) Methanol(2) 79.84 -31.19 813.18 40.73 381.46 346.54 0.2965 Methanol(1) Benzene(2) 40.73 1734.42 183.04 730.09 89.41 1175.41 0.4743 Ethanol(1) Toluene(2) 58.68 1556.45 210.52 106.85 713.57 1147.86 0.5292 Values are those recommended by Gmehling et al., Vapor-Liquid Equilibrium Data Collection, Chemistry Data Series, vol. I, parts la, lb, 2c, and 2e, DECHEMA, Frankfurt/Main, 1981-1988.
Expert Answer:

Related Book For 

Corporate Finance Core Principles And Applications
ISBN: 9781260571127
6th Edition
Authors: Stephen Ross, Randolph Westerfield, Jeffrey Jaffe, Bradford Jordan
Posted Date:
Students also viewed these finance questions
-
Masterson, Inc., has 4 million shares of common stock outstanding. The current share price is $70, and the book value per share is $9. The company also has two bond issues outstanding. The first bond...
-
Dinklage Corp. has 8 million shares of common stock outstanding. The current share price is $74, and the book value per share is $7. The company also has two bond issues outstanding. The first bond...
-
Date 1 July 2019 1 June 2020 30 June 2020 1 July 2020 1 July 2020 30 June 2021 1 July 2021 Particulars (???) (???) (To record acquisition of delivery truck) (???) (???) (???) (To record minor repair...
-
Bathurst Company began April with inventory of $200,000. The business made net purchases of $600,000 and had net sales of $800,000 before a fire destroyed the company's inventory. For the past...
-
(a) From the galvanic series (Table 17.2), cite three metals or alloys that may be used to galvanically protect 304 stainless steel in the active state.
-
Ormet Primary Aluminum Corporation, operated an aluminum smelter plant in Hannibal, Ohio. The facility ceased production in October 2013 in order to liquidate its assets after filing for bankruptcy...
-
Zebra Imaginarium, a retail business, had the following cash receipts during December 20--. The sales tax is 6%. Dec. 1 Received payment on account from Michael Anderson, $1,360. 2 Received payment...
-
Float is defined as the difference between the balance shown on the books and the balance in the bank account. A lag often occurs between the time receipts and disbursements are recorded on the...
-
Question 2: In the November 2019 the price of pork in China increased by more than 100% as a result of the African swine fever crisis which has destroyed half of the pigs in the country. Source: Zou...
-
The Bill of Rights protects individuals against types of interference by the federal government only. (True/False)
-
When do both federal and state courts have the power to hear a case?
-
The purposes of discovery include a. saving time. b. narrowing the issues. c. preventing surprises at trial. d. all of the above.
-
Police officer Malik arrests John on suspicion of embezzlement. Malik advises John of his rights. He informs John a. that John has the right to remain silent. b. that John has the right to consult...
-
During a professional hockey game, Derek, a player for the Devils, collides with Alexei, a player for the Bruins, and falls, hitting his head hard against the ice. Dazed, Derek tells his coach that...
-
What are the information standards used by the COBIT system, with explanation?
-
Federated Shipping, a competing overnight delivery service, informs the customer in Problem 65 that they would ship the 5-pound package for $29.95 and the 20-pound package for $59.20. (A) If...
-
In the previous problem, suppose the fixed asset actually qualifies for 100 percent bonus depreciation. All the other facts are the same. What is the new NPV? Data from Previous Problem Coris Meats...
-
In the previous problem, if the SEC filing fee and associated administrative expenses of the offering are $1.3 million, how many shares need to be sold? Data from Previous Problem The Trafford...
-
Suppose the spot exchange rate for the Canadian dollar is Can$1.29 and the six-month forward rate is Can$1.34. a. Which is worth more, a U.S. dollar or a Canadian dollar? b. Assuming absolute PPP...
-
Consider the homogeneous bar, of negligible cross section, mass \(M=1.333 \mathrm{~kg}\) and length \(L=60.0 \mathrm{~cm}\) in Fig.12.19 . It is constrained to rotate in a vertical plane about its...
-
A projectile of mass \(m=2.5 \mathrm{~kg}\) is shot tangentially (see Fig.12.19) at the edge of a ring having radius \(R=50 \mathrm{~cm}\) and whose mass is equal (within measurement errors) to that...
-
A homogeneous disk, having mass \(M=2.50 \mathrm{~kg}\) and radius \(R=15.0 \mathrm{~cm}\), can rotate, without friction, about a fixed horizontal axis, passing through the point \(\mathrm{O}\) of...

Study smarter with the SolutionInn App


