Do the following calculations and record the results on your Data Sheet. The numbers in parentheses...
Fantastic news! We've Found the answer you've been seeking!
Question:

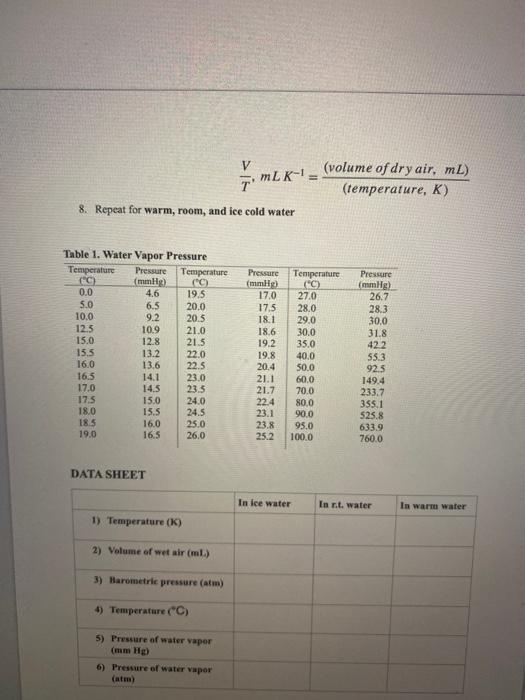


Transcribed Image Text:
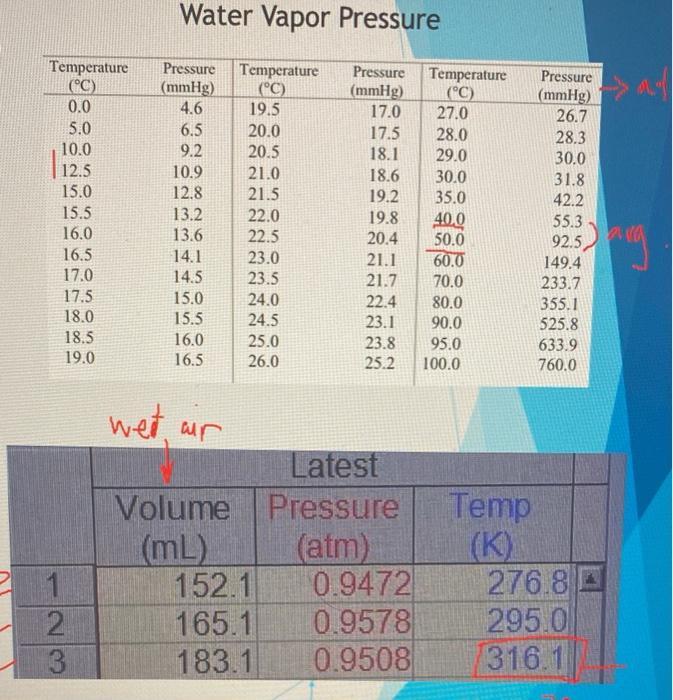
Do the following calculations and record the results on your Data Sheet. The numbers in parentheses refer to the numbered liner on the Data Sheet. 1. Find the volume of wet air. The volume of wet air is the same as the sum of the volume of the flask and the added volume of syringe. This volume is recorded on your Data Sheet (2). 2. Convert the Kelvin temperature (K) to an equivalent temperature in Celsius (°C), using Bauation 1. K- °C+ 273.2 (Eq. 1) Record this temperature on your Data Sheet (4). 3. Find the water vapor pressure at this temperature in the Table 1. If you cannot find the temperature which you look for in the table, use the water vaper pressure at the nearest temperature. Record this water vapor pressure on your Data Sheet (5). 4. Covert the water vapor pressure (mm Hg) to an equivalent pressure in atm, using Equation 2. 1 atm = 760 mm Hg (Eq. 2) Record this water vapor pressure on your Data Sheet (6). 5. Find the pressure of the dry air. Subtract the water vapor pressure (6) from the barometric pressure (3). Record this pressure on your Data Sheet (7). 6. Find the volume of dry air. Multiply the volume of the wet air (2) by the pressure of the dry air (7), divided by the barometric pressure (3), using Equation 3. (Pressure of dry air, atm) (barometric pressure, atm) volume of dry air, ml = (volume of wet air, mL) (Eq 3) Record this volume on your Data Sheet (8) 7. Find the volume-to-termperature ratio (V/T), using Equation 4., Divide the volume of dry air (8) by the Kelvin temperature (1). (volume of dry air, mL) (temperature, K) mLK-= %3D 8. Repeat for warm, room, and ice cold water Table 1. Water Vapor Pressure Temperature C) 0.0 5.0 10.0 Pressure (mmHg) 4.6 Temperature C) 19.5 20.0 20.5 21.0 21.5 22.0 Pressure Temperature ("C) 27.0 28.0 29.0 30.0 35.0 40.0 (mmig) 17.0 Pressure (mmHg) 26.7 6.5 9.2 17.5 18.1 28.3 30.0 31.8 422 55.3 92.5 149.4 233.7 355,1 525.8 633.9 760.0 12.5 15.0 10.9 18.6 12.8 15.5 16.0 16.5 17.0 17.5 18.0 18.5 19.0 19.2 19.8 20.4 21.1 21.7 22.4 23.1 23.8 25.2 13.2 13.6 14.1 14.5 22.5 50.0 23.0 23.5 24.0 24.5 25.0 60.0 70.0 15.0 80.0 15.5 16.0 16.5 90.0 95.0 100.0 26.0 DATA SHEET In lce water In r.t. water In warm water 1) Temperature (K) 2) Volume of wet air (ml.) 3) Barometric pressure (atm) 4) Temperature C) 5) Pressure of water vapor (mm Hg) 6) Pressure ef water vapor (atm) DATA SHEET In ice water In r.t. water In warm water 1) Temperature (K) 2) Volume of wet air (mL) 3) Barometric pressure (atm) 4) Temperature (°C) 5) Pressure of water vapor (mm Hg) 6) Pressure of water vapor (atm) 7) Pressure of dry air (atm) 8) Volume of dry air (mL) 9) V/T (mL/K) Calculations (Show all your work. Use additional paper if necessary) Water Vapor Pressure Temperature (C) 0.0 Pressure Temperature (°C) 19.5 20.0 20.5 Pressure Temperature (C) 27.0 Pressure (mmHg) 4.6 (mmHg) 17.0 (mmHg) 26.7 5.0 6.5 17.5 28.0 29.0 28.3 10.0 9.2 18.1 30.0 12.5 15.0 10.9 21.0 18.6 30.0 31.8 42.2 12.8 21.5 19.2 35.0 15.5 13.2 13.6 22.0 22.5 23.0 23.5 24.0 19.8 20.4 21.1 21.7 40.0 50.0 60.0 55.3 16.0 16.5 92.5 14.1 149.4 17.0 14.5 70.0 233.7 17.5 15.0 22.4 80.0 355.1 18.0 15.5 24.5 23.1 90.0 95.0 525.8 18.5 16.0 25.0 23.8 633.9 19.0 16.5 26.0 25.2 100.0 760.0 wet, ur Latest Volume Pressure (atm) 0.9472 0.9578 0.9508 Temp (K) 276.8 A 295.0 316.1 (mL) 152.1 1 2. 165.1 3 183.1 • Plot temperature (Data Sheet line #1) and volume of dry air (Data Sheet line #8) on graph paper or Excel and put this in your lab report > Use this data and the data from Data Sheet #9 to explain if we can prove Charles's Law (write in your "conclusion" in experimental discussion) Do the following calculations and record the results on your Data Sheet. The numbers in parentheses refer to the numbered liner on the Data Sheet. 1. Find the volume of wet air. The volume of wet air is the same as the sum of the volume of the flask and the added volume of syringe. This volume is recorded on your Data Sheet (2). 2. Convert the Kelvin temperature (K) to an equivalent temperature in Celsius (°C), using Bauation 1. K- °C+ 273.2 (Eq. 1) Record this temperature on your Data Sheet (4). 3. Find the water vapor pressure at this temperature in the Table 1. If you cannot find the temperature which you look for in the table, use the water vaper pressure at the nearest temperature. Record this water vapor pressure on your Data Sheet (5). 4. Covert the water vapor pressure (mm Hg) to an equivalent pressure in atm, using Equation 2. 1 atm = 760 mm Hg (Eq. 2) Record this water vapor pressure on your Data Sheet (6). 5. Find the pressure of the dry air. Subtract the water vapor pressure (6) from the barometric pressure (3). Record this pressure on your Data Sheet (7). 6. Find the volume of dry air. Multiply the volume of the wet air (2) by the pressure of the dry air (7), divided by the barometric pressure (3), using Equation 3. (Pressure of dry air, atm) (barometric pressure, atm) volume of dry air, ml = (volume of wet air, mL) (Eq 3) Record this volume on your Data Sheet (8) 7. Find the volume-to-termperature ratio (V/T), using Equation 4., Divide the volume of dry air (8) by the Kelvin temperature (1). (volume of dry air, mL) (temperature, K) mLK-= %3D 8. Repeat for warm, room, and ice cold water Table 1. Water Vapor Pressure Temperature C) 0.0 5.0 10.0 Pressure (mmHg) 4.6 Temperature C) 19.5 20.0 20.5 21.0 21.5 22.0 Pressure Temperature ("C) 27.0 28.0 29.0 30.0 35.0 40.0 (mmig) 17.0 Pressure (mmHg) 26.7 6.5 9.2 17.5 18.1 28.3 30.0 31.8 422 55.3 92.5 149.4 233.7 355,1 525.8 633.9 760.0 12.5 15.0 10.9 18.6 12.8 15.5 16.0 16.5 17.0 17.5 18.0 18.5 19.0 19.2 19.8 20.4 21.1 21.7 22.4 23.1 23.8 25.2 13.2 13.6 14.1 14.5 22.5 50.0 23.0 23.5 24.0 24.5 25.0 60.0 70.0 15.0 80.0 15.5 16.0 16.5 90.0 95.0 100.0 26.0 DATA SHEET In lce water In r.t. water In warm water 1) Temperature (K) 2) Volume of wet air (ml.) 3) Barometric pressure (atm) 4) Temperature C) 5) Pressure of water vapor (mm Hg) 6) Pressure ef water vapor (atm) DATA SHEET In ice water In r.t. water In warm water 1) Temperature (K) 2) Volume of wet air (mL) 3) Barometric pressure (atm) 4) Temperature (°C) 5) Pressure of water vapor (mm Hg) 6) Pressure of water vapor (atm) 7) Pressure of dry air (atm) 8) Volume of dry air (mL) 9) V/T (mL/K) Calculations (Show all your work. Use additional paper if necessary) Water Vapor Pressure Temperature (C) 0.0 Pressure Temperature (°C) 19.5 20.0 20.5 Pressure Temperature (C) 27.0 Pressure (mmHg) 4.6 (mmHg) 17.0 (mmHg) 26.7 5.0 6.5 17.5 28.0 29.0 28.3 10.0 9.2 18.1 30.0 12.5 15.0 10.9 21.0 18.6 30.0 31.8 42.2 12.8 21.5 19.2 35.0 15.5 13.2 13.6 22.0 22.5 23.0 23.5 24.0 19.8 20.4 21.1 21.7 40.0 50.0 60.0 55.3 16.0 16.5 92.5 14.1 149.4 17.0 14.5 70.0 233.7 17.5 15.0 22.4 80.0 355.1 18.0 15.5 24.5 23.1 90.0 95.0 525.8 18.5 16.0 25.0 23.8 633.9 19.0 16.5 26.0 25.2 100.0 760.0 wet, ur Latest Volume Pressure (atm) 0.9472 0.9578 0.9508 Temp (K) 276.8 A 295.0 316.1 (mL) 152.1 1 2. 165.1 3 183.1 • Plot temperature (Data Sheet line #1) and volume of dry air (Data Sheet line #8) on graph paper or Excel and put this in your lab report > Use this data and the data from Data Sheet #9 to explain if we can prove Charles's Law (write in your "conclusion" in experimental discussion)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Water in an open dish exposed to dry air at 25oC is found to vaporize at a constant rate of 0.04 g/h-cm2. Assuming the water surface to be at the wet-bulb temperature of ll.0C, calculate the...
-
Water at a gauge pressure of 3.8 atm at street level flows into an office building at a speed of 0.60m/s through a pipe 5.0cm in diameter, the pipe tapers down to 2.6cm in diameter by the top floor,...
-
Water is boiling at 1 atm pressure in a stainless steel pan on an electric range. It is observed that 2 kg of liquid water evaporates in 30 min. The rate of heat transfer to the water is (a) 2.51 kW...
-
Find the smallest positive angle and the smallest negative angle (numerically) coterminal with but not equal to the given angle. 47.0
-
Twenty-five slips of paper, numbered 1, 2, 3, ... , 25, are placed in a box. If Amy draws six of these slips, without replacement, what is the probability that (a) The second smallest number drawn is...
-
As of May 31, Year 1, the bank statement showed an ending balance of $26,100. The unadjusted Cash account balance was $27,350. The following information is available: 1. Deposit in transit: $6,981....
-
Use information from Section 6.7 to estimate which form of electromagnetic radiation is the lowest energy ionizing radiation. Data from section 6.7 When we first introduced the concept of the...
-
Consider the unadjusted trial balance of Princess, Inc., at August 31, 2012, and the related month-end adjustment data. Adjustment data at August 31, 2012, include the following: a. Accrued...
-
Alberto plans to apply for a car loan. When should he check his credit report? Group of answer choices 1 month before applying for the loan Around 3 to 6 months before applying for the loan 1 week...
-
The financial statements at the end of Wolverine Realty's first month of operations are as follows: Instructions By analyzing the interrelationships among the four financial statements, determine the...
-
To find any information on the Internet, we enter specific keywords into a search engine. What happens next? A. The search engine looks up the required data from the giant database that has all the...
-
The case focuses on QANTAS, and the role of Olivia Wirth (executive of Government and Corporate Affairs) who has to respond to the company's failed and publicly scrutinised social media contest...
-
As you might know, during 2021 we faced a major issue in our Operations/Supply Chain: Global Supply Chain collapse that affected several industries and customers Questions that need to be answered 1....
-
Submit a draft of Section I (Introduction) based on the strategic plan of the agency. Note that you will be writing about the Acme Health Care Agency PDF This draft should be as detailed as possible...
-
Which is the image of following mapping? 2 0-5 4 3 7
-
Every organization should develop a portfolio management framework that fits its business and includes the following elements: 1. a project review board, 2. structured, consistent collection and...
-
The authors describe how two Amazon workers managed to organize a union in their warehouse. The company spent more than $4.3 million on anti-union consultants across the US in the same period....
-
Q:1 Take any product or service offered in Pakistan and apply all determinents of customer Perceived value ?
-
The article "Organic Recycling for Soil Quality Conservation in a Sub-Tropical Plateau Region" (K. Chakrabarti, B. Sarkar, et al., J. Agronomy and Crop Science, 2000:137-142) reports an experiment in...
-
In the article "Occurrence and Distribution of Ammonium in Iowa Groundwater" (K. Schilling, Water Environment Research, 2002:177-186), ammonium concentrations (in mg/L) were measured at a large...
-
The thickness, in mm, of metal washers is measured on samples of size 5. The following table presents the means, ranges, and standard deviations for 20 consecutive samples. The means are = 2.505, =...
-
The plaintiff [Christine Parent] leased an automobile from Amity Autoworld, Ltd. (hereafter Amity) [in Amityville, New York] in January 2002. Amity sold all its Toyota automobile franchise assets * *...
-
PT China, LLC, joined with PT Korea, LLC, to form PT Holdings. Harrison Wang and Michael Kim, representing the two entities, were to be the managers. The agreements prohibited them from engaging in...
-
The Communications Act of 1934 established a system of limited-term broadcast licenses subject to various conditions * * * . * * * [These conditions include] the indecency banthe statutory...

Study smarter with the SolutionInn App


