Question: Define Chemical equilibrium. Determine the equilibrium constant (kc) for the synthesis of PCls from PCI3 and Cl2 (PCI3 +Cl2 PCI5).
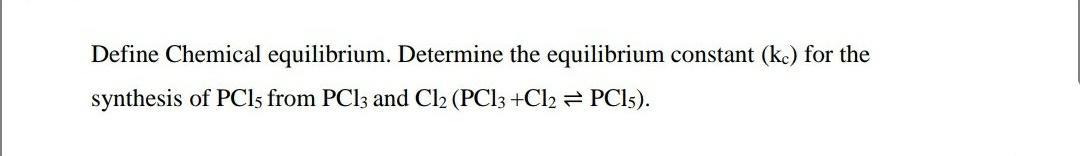
Define Chemical equilibrium. Determine the equilibrium constant (kc) for the synthesis of PCls from PCI3 and Cl2 (PCI3 +Cl2 PCI5).
Step by Step Solution
★★★★★
3.34 Rating (157 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Solution Chemical equilibrium When the rate of the forward react... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock


