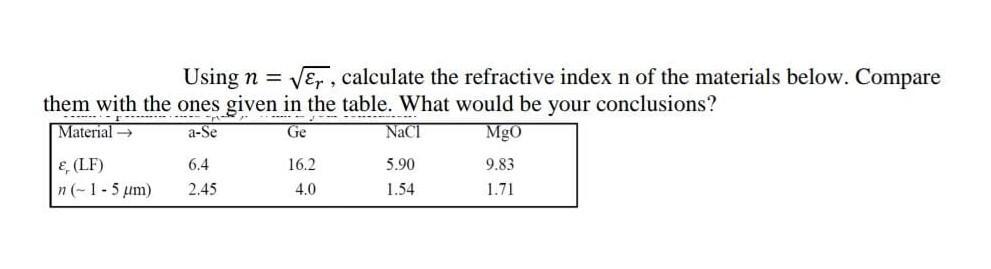
Using n = , calculate the refractive index n of the materials below. Compare them with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Using n = √√, calculate the refractive index n of the materials below. Compare them with the ones given in the table. What would be your conclusions? --------- F ...pat Material → a-Se Ge NaCl MgO &, (LF) n (- 1 - 5 um) 6.4 2.45 16.2 4.0 5.90 1.54 9.83 1.71 Using n = √√, calculate the refractive index n of the materials below. Compare them with the ones given in the table. What would be your conclusions? --------- F ...pat Material → a-Se Ge NaCl MgO &, (LF) n (- 1 - 5 um) 6.4 2.45 16.2 4.0 5.90 1.54 9.83 1.71
Expert Answer:
Answer rating: 100% (QA)
Answer Material rLF n15 m Er N In N a Se 64 245 253 008 Ge 16... View the full answer

Related Book For 

Probability and Statistics for Engineering and the Sciences
ISBN: 978-1305251809
9th edition
Authors: Jay L. Devore
Posted Date:
Students also viewed these mechanical engineering questions
-
What would be your conclusions for these various acceptance rates? A cost-benefit analysis is a systematic comparison of the costs and benefits of a proposed action. Only if the benefits exceed the...
-
What would be your reaction if you were told you were not hired based on your responses to this questionnaire?
-
What would be your strategy for changing and creating an entrepreneurial culture in a large no entrepreneurial culture in a large, nonentrepreneurial firm? Is it possible? Why or why not?
-
What is a time series? Explain the four characteristics that time series may exhibit and provide some practical examples.
-
Personal Care Products Company is considering an investment in one of two new product lines. The investment required for either product line is $550,000. The net cash flows associated with each...
-
Fall-Line, Inc. is a Great Falls, Montana, manufacturer of a variety of downhill skis. Fall-line is considering four locations for a new plant: As Pen Colorado; Medicine Lodge Kansas; Broken Bow,...
-
For the following separate bond issues, assume that the bonds are sold on January 1, 2020, interest is paid semiannually on July 1 and December 31, and the bond term is 5 years. Required Complete the...
-
(Second Year of Depreciation Difference, Two Differences, Single Rate, Extraordinary Item) The following information has been obtained for the Gocker Corporation. 1. Prior to 2010, taxable income and...
-
You need to calculate the cost of debt for Tesla. Find the yield to maturity for each of Tesla's bonds. What is the weighted average cost of debt for Tesla using the book value weights and the...
-
Compare PengAtlas Maps 2.1 (Top Merchandise Importers and Exporters), 2.2 (Top Service Importers and Exporters), and 2.3 (FDI Inflows and Outflows) once again and note the BRIC countries that are...
-
Suppose a certain drug test is 80% sensitive, that is, the test will correctly identify a drug user as testing positive 80% of the time, and 92% specific, that is, the test will correctly identify a...
-
John is voluntarily turned over to the custody of the state of Florida. John is autistic. He is placed in a foster home with five (5) other children. John's autism requires specialized care which he...
-
Casey Anthony Case Casey Anthony Case Study Focus on Evidence in Case Please type the answers below each point. Please let me know if you have any questions with this. Read the article about some of...
-
in 1985, a and his friend, z, created In 1995, a man and his friend created a corporation. The man owned 55% of the stock, and the friend owned 45% of the stock. When the man died in 2005, he left...
-
When a multi-stage booster is fired into space, the mass of the booster changes as the fuel is burned during flight. Discuss how the change in mass influences the acceleration of the booster. When...
-
Last year, the State Board of Education in the State of Jefferson made some changes in the prescribed history curriculum in its public schools. In the "New History Curriculum," all American history...
-
How many distinct monochlorinated products, including stereoisomers, can result when the alkane below is heated in the presence of Cl 2 ? I know the correct answer is 7, but can anyone draw all the...
-
Test your confidence in the following Project Decisions: SI. # Question 01 02 03 04 05 06 07 08 09 10 How many years did it take to construct the largest Egyptian Pyramid- Pyramid of Cheops? When was...
-
The accompanying two-way frequency table appears in the article "Marijuana Use in College" (Youth and Society, 1979: 323-334). Each of 445 college students was classified according to both frequency...
-
The accompanying table gives sample means and standard deviations, each based on n = 6 observations of the refractive index of fiber-optic cable. Construct a control chart, and comment on its...
-
Write a general rule for E(X - c) where c is a constant. What happens when c = , the expected value of X?
-
7. ROLE REVERSAL Write a short-answer question that deals with an issue involving crime in cyberspace.
-
Question: A police department supplied its officers with pagers for text-messaging. The department did not have an official policy on text-messaging, but its e-mail policy stated that: "The use of...
-
Question: In 1572, during the reign of Queen Elizabeth I of England, a patent application was filed for a knife with a bone handle rather than a wooden one. Would this patent be granted under current...

Study smarter with the SolutionInn App


