DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your...
Fantastic news! We've Found the answer you've been seeking!
Question:

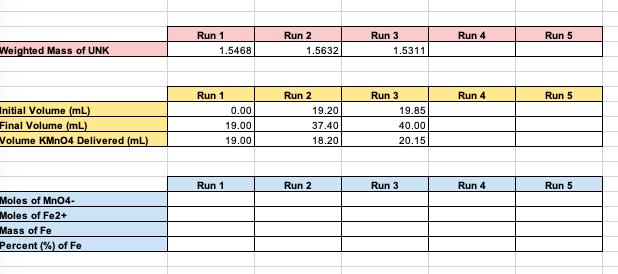
Transcribed Image Text:
DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your professor and record the unknown number in your notebook. Weigh out 3-4 samples of between 1.5000g and 2.0000g ferrous ammonium sulfate unknown and place in 250 mL flasks. Add 15 mL of distilled water and 1 mL of concentrated HCI to each sample. Swirl to dissolve sample completely. Gently warm on a hot plate at your bench. Wash down the sides of the flask with some distilled water. Add 0.2 M Potassium Permanganate solution (not your standardized solution) dropwise until a distinct yellow color forms. (should be about 2 drops) Carry out the following procedure individually on each sample. Add 25 mL of the Zimmermann-Reinhardt reagent and 300 mL of distilled water. Titrate immediately with your standardized KMnO4 solution to the first faint pink color that persists for 15 to 30 seconds. Do not titrate rapidly at any time. The pink color results from excess MnO4, indicating that all Fe²+ has been oxidized to Fe³+, Weighted Mass of UNK Initial Volume (mL) Final Volume (mL) Volume KMnO4 Delivered (mL) Moles of MnO4- Moles of Fe2+ Mass of Fe Percent (%) of Fe Run 1 1.5468 0.00 19.00 19.00 Run 1 Run 1 Run 2 1.5632 19.20 37.40 18.20 Run 2 Run 2 Run 3 1.5311 19.85 40.00 20.15 Run 3 Run 3 Run 4 Run 4 Run 4 Run 5 Run 5 Run 5 DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your professor and record the unknown number in your notebook. Weigh out 3-4 samples of between 1.5000g and 2.0000g ferrous ammonium sulfate unknown and place in 250 mL flasks. Add 15 mL of distilled water and 1 mL of concentrated HCI to each sample. Swirl to dissolve sample completely. Gently warm on a hot plate at your bench. Wash down the sides of the flask with some distilled water. Add 0.2 M Potassium Permanganate solution (not your standardized solution) dropwise until a distinct yellow color forms. (should be about 2 drops) Carry out the following procedure individually on each sample. Add 25 mL of the Zimmermann-Reinhardt reagent and 300 mL of distilled water. Titrate immediately with your standardized KMnO4 solution to the first faint pink color that persists for 15 to 30 seconds. Do not titrate rapidly at any time. The pink color results from excess MnO4, indicating that all Fe²+ has been oxidized to Fe³+, Weighted Mass of UNK Initial Volume (mL) Final Volume (mL) Volume KMnO4 Delivered (mL) Moles of MnO4- Moles of Fe2+ Mass of Fe Percent (%) of Fe Run 1 1.5468 0.00 19.00 19.00 Run 1 Run 1 Run 2 1.5632 19.20 37.40 18.20 Run 2 Run 2 Run 3 1.5311 19.85 40.00 20.15 Run 3 Run 3 Run 4 Run 4 Run 4 Run 5 Run 5 Run 5
Expert Answer:
Answer rating: 100% (QA)
Molasity of Initial V ml 010 1920 1985 Final V ml 190 3740 4000 V 190 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
A dilute solution of potassium permanganate in water at 25C was prepared. The solution was in a horizontal tube of length 10 cm, and at first there was a linear gradation of intensity of the purple...
-
Potassium sulfate solution reacts with barium bromide solution to produce a precipitate of barium sulfate and a solution of potassium bromide. Write the molecular equation for this reaction. Then...
-
A 35.2-mL, 1.66 M KMnO4 solution is mixed with 16.7mL of 0.892 M KMnO4 solution. Calculate the concentration of the final solution.
-
Susan recently quit working for a local firm and has yet to find a new job. She knows she can maintain her health insurance from her old employer due to COBRA. How much will it likely cost her for...
-
The Shinkansen, the Japanese "bullet" train, runs at high speed from Tokyo to Nagoya. Riding on the Shinkansen, you notice that the frequency of a crossing signal changes markedly as you pass the...
-
Are personality tests useful tools for organizational hiring?
-
How would you define the concepts organizational environment and environmental domain? LO.1
-
Roselle Appliance uses a perpetual inventory system. For its fl at-screen television sets, the January 1 inventory was 3 sets at $600 each. On January 10, Roselle purchased 6 units at $648 each. The...
-
Meir, Benson, and Lau are partners and share income and loss in a 2:3:5 ratio (in percents: Meir, 20%; Benson, 30%; and Lau, 50%). The partnership's capital balances are as follows: Meir, $88,000;...
-
In Integrative Case 10.1, we projected financial statements for Walmart Stores for Years +1 through +5. The data in Chapter 12, Exhibits 12.17 through 12.19 include the actual amounts for 2015 and...
-
Which characteristic is correct for the function f(x)=-2x+3x? O both even and odd O even O neither even nor odd O odd
-
Let f(x) = x+ 3, x20. The inverse of f is Of 1(x)=x - 3 (f (x) = -x-3 f-(x) = x - 3 Of 1(x) = 3 - x
-
Read the articles and please help me to write the whole assignment perfectly including the citations and references (APA Format). Pleaase choose the country and perspective of a particular industry....
-
A light, inextensible cord passes over a frictionless pulley as shown in figure below. One end of the rope is attached to a block, and a force P is applied to the other end. Block A weighs 600 lb and...
-
BASICOT POST DO NOT ASSIST DO NOT POST DO NOT ASSIST DO NOT POST DO NOT ASSIST For filming a physics demonstration about oscillation, an educational video crew attaches a large spring to a very small...
-
Day Mail Order Co. applied the high-low method of cost estimation to customer order data for the first 4 months of the year. What is the estimated variable order-filling cost component per order...
-
Doug's Custom Construction Company is considering three new projects, each requiring an equipment investment of $26,840. Each project will last for 3 years and produce the following net annual cash...
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
An acidic solution containing 0.010 M La3+ is treated with NaOH until La(OH)3 precipitates. At what pH does this occur?
-
Sulfide ion was determined by indirect titration with EDTA. To a solution containing 25.00 mL of 0.04332 M Cu(ClO4)2 plus 15 mL of 1M acetate buffer (pH 4.5) were added 25.00 mL of unknown sulfide...
-
What are the general steps in developing an isocratic separation for reversed-phase chromatography with one organic solvent and temperature as variables?
-
If a forecasted purchase of equipment were to be denominated in foreign currency (FC), how would the change in value of a cash flow hedge of the forecasted transaction be accounted for? AppendixLO1
-
Construct a single lift chart which includes the better of the two CART models, the better of the two C4.5 models, and the neural network model. Which model is preferable over which regions?
-
Explain the floating international monetary system, and identify factors that influence rates of exchange between currencies. AppendixLO1

Study smarter with the SolutionInn App


