Question: Develop a constant-pressure-reactor model using the same chemistry and thermodynamics as in Example 6.1. Using an initial volume of 0.008 m 3 , P =
Develop a constant-pressure-reactor model using the same chemistry and thermodynamics as in Example 6.1. Using an initial volume of 0.008 m3, P = 1 atm and To= 1000 K, (a) find the combustion durations such that the reaction is 99 percent complete. Use ? = 1 and assume the reactor is adiabatic.
b) Study the effect of variation in?P?[0.2 ? 10 atm]?and?T0?[700???1400 K] on combustion durations. Use ? = 1 and assume the reactor is adiabatic.
Assumptions in example 6.1 are as follow:
- One-step global kinetics using the rate parameters for ethane C2H6?(Table 5.1)
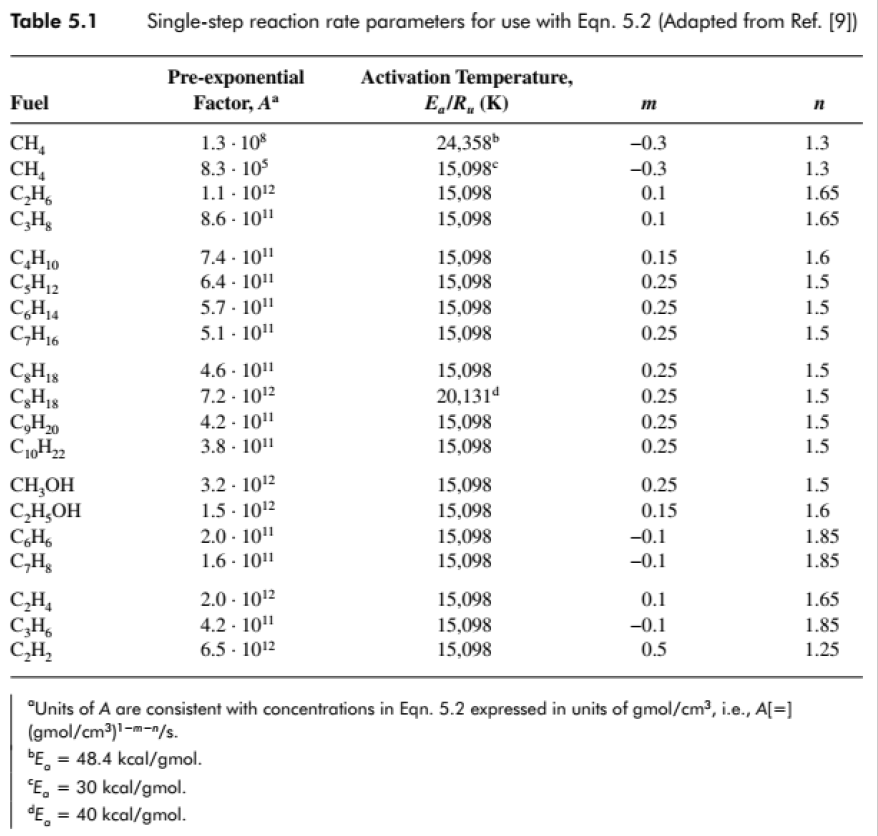
- Fuel, air, and products all have equal molecular weights: MWF= MWOx= MWP= 29
- The specific heats of the fuel, air and products are constants and equal:
- cp,F= cp,Ox= cp,Pr= 1200 J/kgK
- The enthalpy of formation of the air and products are zero, and that of the fuel is
- 4*107j/kg
- The stoichiometric air-fuel ratio is 16.0 and restrict combustion to stoichiometric or lean conditions.excel must be formatted as following
![rate parameters for use with Eqn. 5.2 (Adapted from Ref. [9]) Pre-exponential](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/11/63625fe92af41_46563625fe92ac27.jpg)
?
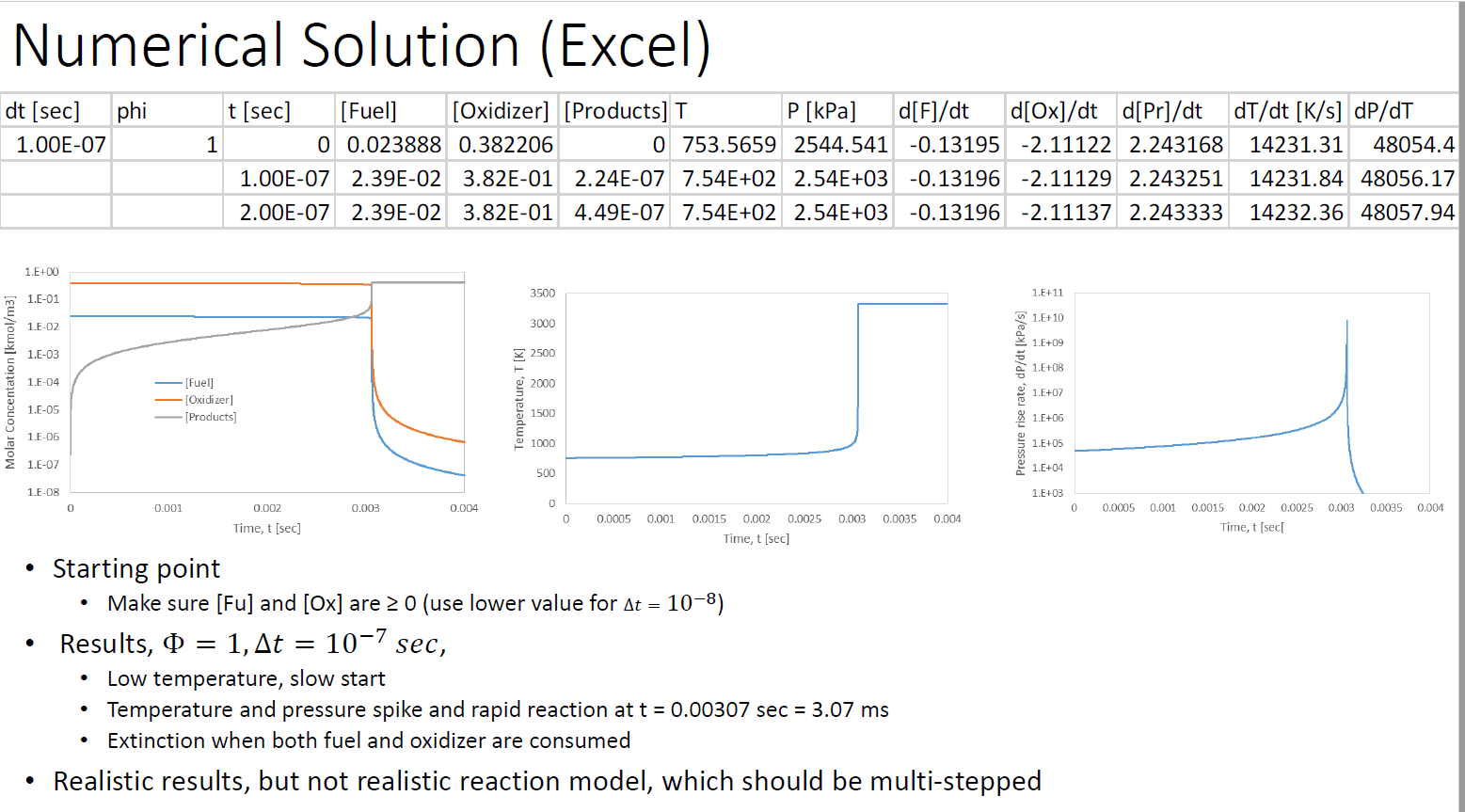
Table 5.1 Fuel CH CHA CH C3H8 CH0 CH12 CH4 CH16 C8H18 C8H18 CH0 C10H2 CHOH CHOH C6H6 CH8 CH C3H6 CH Single-step reaction rate parameters for use with Eqn. 5.2 (Adapted from Ref. [9]) Pre-exponential Factor, A 1.3.108 8.3 105 1.1 - 102 8.6-101 7.4-10 6.4. 101 5.7.10 5.1 10 4.6-1011 7.2.102 4.2.10 3.8 101 3.2 102 1.5-102 2.0-10 1.6-101 2.0-102 4.2.101 6.5 - 102 Activation Temperature, E/R (K) bE = 48.4 kcal/gmol. E = 30 kcal/gmol. E = 40 kcal/gmol. 24,358b 15,098 15,098 15,098 15,098 15,098 15,098 15,098 15,098 20,1314 15,098 15,098 15,098 15,098 15,098 15,098 15,098 15,098 15,098 m -0.3 -0.3 0.1 0.1 0.15 0.25 0.25 0.25 0.25 0.25 0.25 0.25 0.25 0.15 -0.1 -0.1 0.1 -0.1 0.5 "Units of A are consistent with concentrations in Eqn. 5.2 expressed in units of gmol/cm, i.e., A[=] (gmol/cm)1-m-n/s. n 1.3 1.3 1.65 1.65 1.6 1.5 1.5 1.5 1.5 1.5 1.5 1.5 1.5 1.6 1.85 1.85 1.65 1.85 1.25 Numerical Solution (Excel) phi P [kPa] d[F]/dt d[Ox]/dt d[Pr]/dt dT/dt [K/s] dp/dT [Fuel] [Oxidizer] [Products] T 0 0.023888 0.382206 0 753.5659 2544.541 -0.13195 -2.11122 2.243168 14231.31 48054.4 1.00E-07 2.39E-02 3.82E-01 2.24E-07 7.54E+02 2.54E+03 -0.13196 -2.11129 2.243251 14231.84 48056.17 2.00E-07 2.39E-02 3.82E-01 4.49E-07 7.54E+02 2.54E+03 -0.13196 -2.11137 2.243333 14232.36 48057.94 dt [sec] 1.00E-07 Molar Concentation [kmol/m3] 1.E+00 1.E-01 1.E-02 1.E-03 1.E-04 1.E-05 1.E-06 1.E-07 1.E-08 0 1 0.001 t [sec] [Fuel] - [Oxidizer] [Products] 0.002 Time, t [sec] Results, = 1, At 0.003 = 0.004 Temperature, T [K] 3500 3000 2500 2000 1500 1000 500 0 0 0.0005 0.001 Starting point Make sure [Fu] and [Ox] are 0 (use lower value for At = 10-8) 10-7 sec, 0.0015 0.002 0.0025 0.003 0.0035 0.004 Time, t [sec] Pressure rise rate, dp/dt [kPa/s] 1.E+11 1.E+10 1.E+09 1.E+08 1.E+07 1.E+06 1.E+05 1.E+04 1.E+03 Low temperature, slow start Temperature and pressure spike and rapid reaction at t = 0.00307 sec = 3.07 ms Extinction when both fuel and oxidizer are consumed Realistic results, but not realistic reaction model, which should be multi-stepped 0 0.0005 0.001 0.0015 0.002 0.0025 0.003 0.0035 0.004 Time, t [sec[ Dependence on , = 0.5 1.E+00 1.E-01 Molar Concentation [kmol/m3] 1.E-02 1.E-03 1.F-04 1.E-05 1.E-06 1.E-07 0 = 0.1 1.E+00 1.E-01 1.E-02 1.E-03 1.E-04 1.E-05 1.E-06 1.E-07 1.E-08 0 [Fuel] -[Oxidizer] [Products] 0.001 0.001 0.002 Time, t [sec] [Fuel] [Oxidizer] [Products] 0.002 Time, t [sec] 0.003 0.003 0.004 0.004 Temperature, T [K] 3500 3000 2500 2000 1500 1000 500 Temperature, T [K] 0 0 3500 3000 2500 2000 1500 1000 500 0 0 At = 10-7 sec 0.0005 0.001 0.0015 0.002 0.0025 Time, t [sec] 0.0005 0.001 0.003 0.0035 0.004 0.0015 0.002 0.0025 0.003 0.0035 0.004 Time, t [sec] Pressure rise rate, dp/dt [kPa/s] Pressure rise rate, dp/dt [kPa/s] 1.E+11 1.E+10 1.E+09 1.E+08 1.E+07 1.E+06 1.E+05 1.E+04 1.E+03 1.E+11 1.E+10 . 1.E+09 1.E+08 1.E+07 1.E+06 1.E+05 1.E+04 1.E+03 0 0.0005 0.001 0.0015 0.002 0 0.0005 0.001 As before the reaction is "delayed" Initial [Fuel] is decreases Due to excess air, the mixture takes longer to heat up, and reaches lower temperatures and pressures Lower temperatures delay reaction to 3.12 and 3.53 ms (3.07 ms for $ = 1) 0.0025 0.003 0.0035 0.004 Time, t [sec[ 0.0015 0.002 0.0025 0.003 0.0035 0.004 Time, t [sec[
Step by Step Solution
3.64 Rating (177 Votes )
There are 3 Steps involved in it
To develop a constantpressurereactor model and analyze the combustion duration well follow these steps Step 1 Define the Reaction Model Chemical React... View full answer

Get step-by-step solutions from verified subject matter experts


