Question: Directions: Analyze the flow chart carefully and answer the missing parts based on the words given at the box below. 4. Hydrogen Bonding Van
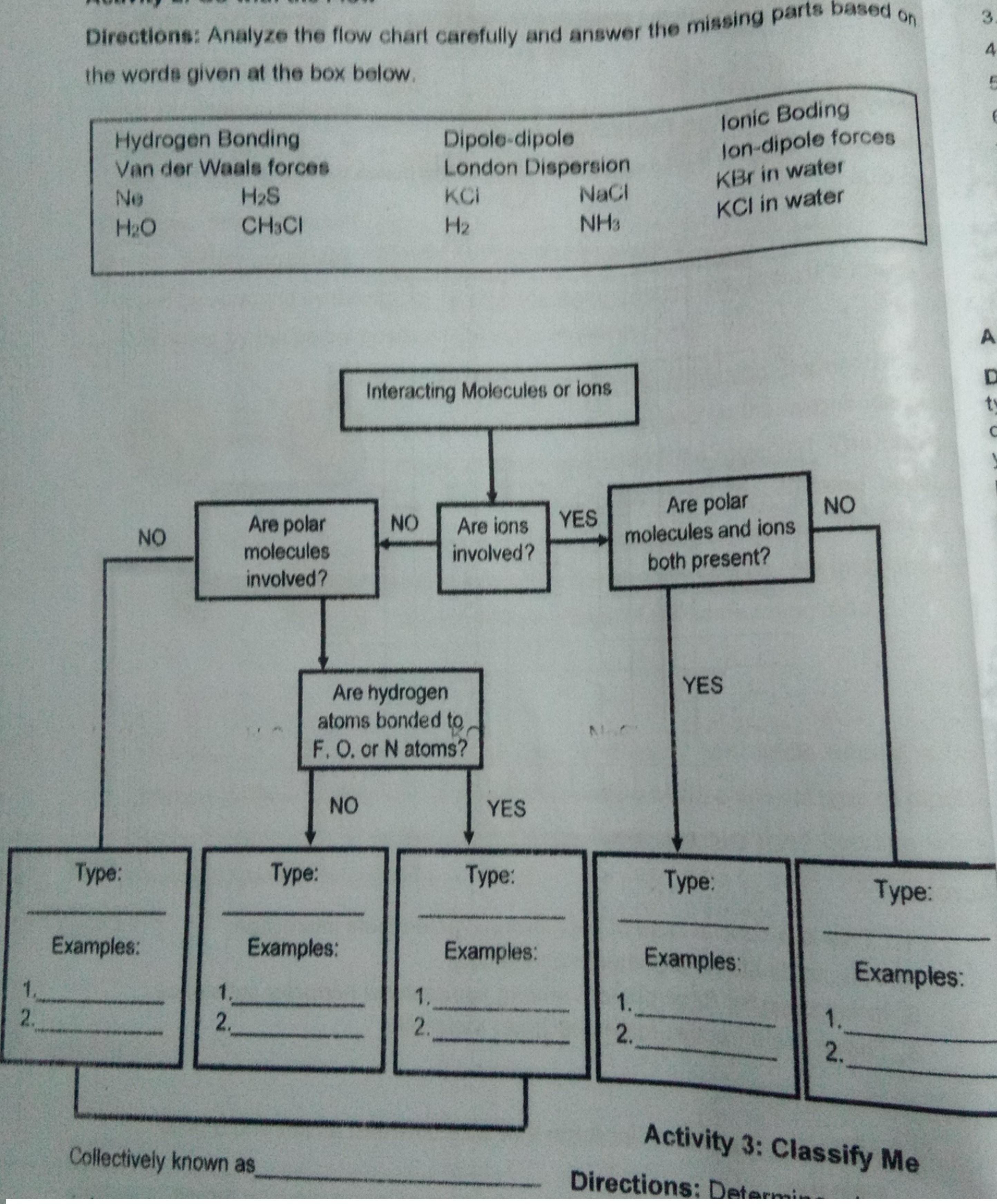
Directions: Analyze the flow chart carefully and answer the missing parts based on the words given at the box below. 4. Hydrogen Bonding Van der Waals forces H2S Dipole-dipole London Dispersion NaCl lonic Boding lon-dipole forces KBr in water KCI in water Ne KCI H2O CH3CI H2 NH3 Interacting Molecules or ions tv Are polar NO Are polar molecules Are ions YES involved? NO NO molecules and ions both present? involved? YES Are hydrogen atoms bonded to, F. O. or N atoms? NO YES : e: Type: p: e: Examples: Examples: Examples: Examples: Examples: 1. 2. 1. 1. 1. 2. 2. 2. 2. Activity 3: Classify Me Collectively known as Directions: Determi
Step by Step Solution
3.43 Rating (153 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


