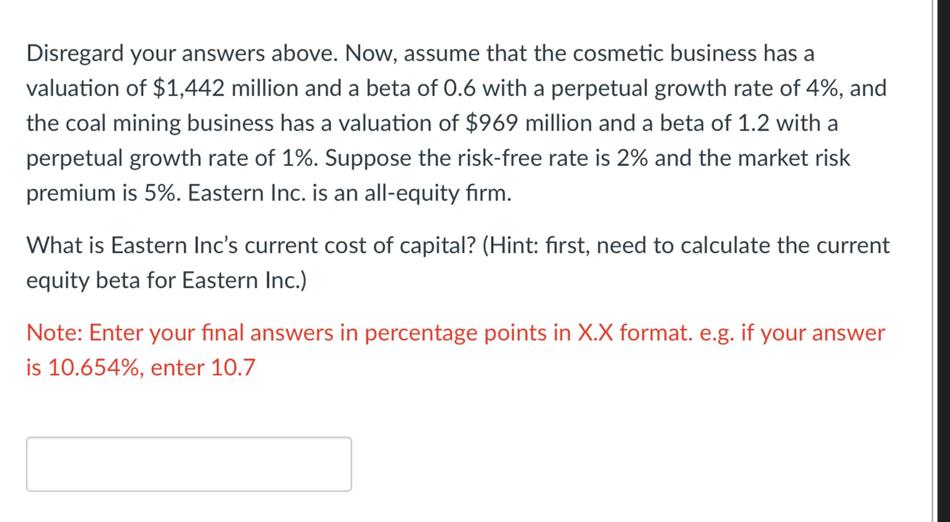
Disregard your answers above. Now, assume that the cosmetic business has a valuation of $1,442 million...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Disregard your answers above. Now, assume that the cosmetic business has a valuation of $1,442 million and a beta of 0.6 with a perpetual growth rate of 4%, and the coal mining business has a valuation of $969 million and a beta of 1.2 with a perpetual growth rate of 1%. Suppose the risk-free rate is 2% and the market risk premium is 5%. Eastern Inc. is an all-equity firm. What is Eastern Inc's current cost of capital? (Hint: first, need to calculate the current equity beta for Eastern Inc.) Note: Enter your final answers in percentage points in X.X format. e.g. if your answer is 10.654%, enter 10.7 Disregard your answers above. Now, assume that the cosmetic business has a valuation of $1,442 million and a beta of 0.6 with a perpetual growth rate of 4%, and the coal mining business has a valuation of $969 million and a beta of 1.2 with a perpetual growth rate of 1%. Suppose the risk-free rate is 2% and the market risk premium is 5%. Eastern Inc. is an all-equity firm. What is Eastern Inc's current cost of capital? (Hint: first, need to calculate the current equity beta for Eastern Inc.) Note: Enter your final answers in percentage points in X.X format. e.g. if your answer is 10.654%, enter 10.7
Expert Answer:
Answer rating: 100% (QA)
To calculate Eastern Incs current cost of capital we first need to find its equity beta using the be... View the full answer

Related Book For 

Financial Reporting Financial Statement Analysis and Valuation a strategic perspective
ISBN: 978-1285190907
8th edition
Authors: James M. Wahlen, Stephen P. Baginski, Mark Bradshaw
Posted Date:
Students also viewed these finance questions
-
a. Describe briefly the legal rights and privileges of common stockholders. b. What is free cash flow (FCF)? What is the weighted average cost of capital? What is the free cash flow valuation model?...
-
Last Sale Net Bid Ask Open Int Puts Last Sale Net bid Ask Vol Open Int 16Aug 155.00(1619H155-E) 6.45 0.75 6.95 7.25 9 16Aug 155.00(1619T155-E) 1.18 (0.75) 1.17 1.25 61 4505 16Aug 166.00(1619H160-E)...
-
If the Stock Dividends < 25%, recorded at fair market value. If the Stock Dividends > 25%, recorded at book value. Example1: Velvet Company has 5,000 shares issued and outstanding. Par value is $1;...
-
Discuss how you would develop a profile of an effective leader from the research results of the GLOBE project. Give an example.
-
Use the graph of f and g and their tangent lines at (2, 0) to find f(x)/g(x). im yt y= 1.8(x-2) // f/ 2 0 y=5(x-2)
-
What makes a question bad? Develop three examples of bad questions. Rewrite your examples so they could be judged as good questions.
-
Maypole Industries imports goods from Taiwan and resells them to domestic Canadian markets. Maypole uses a perpetual inventory system. A typical transaction stream follows: Required: 1. Prepare...
-
Reflect on the role of the external context of policy-making considering the recent Supreme Court's decisions related to affirmative action and student' loans. What does this process say about the...
-
Melodic Musical Sales, Inc. is located at 5500 Fourth Avenue, City, ST 98765. The corporation uses the calendar year and accrual basis for both book and tax purposes. It is engaged in the sale of...
-
Snoopy drives a 1000-kg car moving at 10 m/s and brakes to a stop in 5.00 s. What would the average braking force have been?
-
Every week a truckload of springs is delivered to the warehouse you supervise. Every time the springs arrive, you have to measure the strength of 400 springs. You accept the shipment only when there...
-
At \(35^{\circ} \mathrm{C}\), the root-mean-square speed of a certain monatomic ideal gas is \(186 \mathrm{~m} / \mathrm{s}\). (a) What is the mass of each atom? (b) What is the average kinetic...
-
An engineer takes a sample of 5 steel I beams from a batch, and measures the amount they sag under a standard load. The amounts in \(\mathrm{mm}\) are: It is known that the sag is normal( \({ }^{2}\)...
-
A sample of neon gas is at \(100^{\circ} \mathrm{C}\). The mass of a neon atom is \(3.35 \times 10^{-26} \mathrm{~kg}\). (a) What is the average kinetic energy of a neon atom? \((b)\) If the thermal...
-
A manufacturer of toys is employing fifty workers and using fifteen pieces of equipment to assemble toys. Currently, the marginal product of labor is $5 and the marginal product of capital is $25....
-
Draw a graph that plots Short-run Aggregate Supply, Long.Run Aggregate Supply, and Aggregate De- mand. Indicate the equilibrium point on the graph. Then, explain the shifts of the curves and the...
-
You have accepted the engagement of auditing the financial statements of the C. Reis Company, a small manufacturing firm that has been your auditee for several years. Because you were busy writing...
-
Exhibit 8.29 presents the separate financial statements at December 31, 2015, of Prestige Resorts and its 80%-owned subsidiary Booking, Inc. Two years earlier on January 1, 2014, Prestige acquired...
-
a. Use the CAPM to compute the required rate of return on equity capital for Starbucks. b. Compute the weighted-average cost of capital for Starbucks as of the start of Year 1. At the start of Year...
-
Whirlpool manufactures and sells home appliances under various brand names. IBM develops and manufactures computer hardware and offers related technology services. Target operates a chain of general...
-
A vertical belt is moving upward continuously through a liquid bath at a velocity $V$. A film of the liquid adheres to the belt, which tends to drain downward due to gravity. The equilibrium...
-
A slider bearing can be described as one plate moving with a velocity $V$ parallel to a stationary plate, with a viscous lubricant in between the plates. The force applied to the moving plate is $F$,...
-
Acrylic latex paint can be described as a Bingham plastic with a yield stress of $200 \mathrm{dyn} / \mathrm{cm}^{2}$, a limiting viscosity of $50 \mathrm{cP}$, and a density of $0.95 \mathrm{~g} /...

Study smarter with the SolutionInn App


