Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solutino...
Fantastic news! We've Found the answer you've been seeking!
Question:
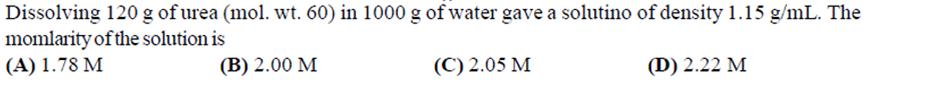

Transcribed Image Text:
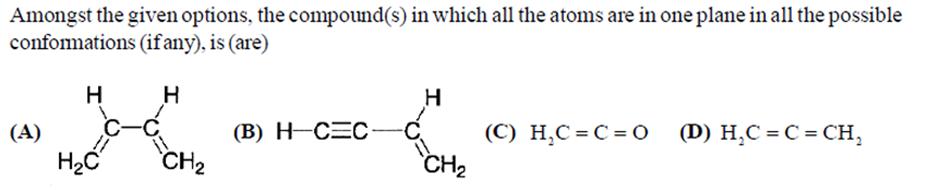
Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solutino of density 1.15 g/mL. The momlarity of the solution is (A) 1.78 M (B) 2.00 M (C) 2.05 M (D) 2.22 M Extraction of metal from the ore casiterite involves (A) carbon reduction of an oxide ore (C) removal of copper (B) self-reduction of a sulphide ore (D) removal of iron impurity Amongst the given options, the compound(s) in which all the atoms are in one plane in all the possible conformations (if any), is (are) (A) H HC C-C H CH (B) H-CEC H -C CH (C) HC=C=O (D) HC=C=CH Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solutino of density 1.15 g/mL. The momlarity of the solution is (A) 1.78 M (B) 2.00 M (C) 2.05 M (D) 2.22 M Extraction of metal from the ore casiterite involves (A) carbon reduction of an oxide ore (C) removal of copper (B) self-reduction of a sulphide ore (D) removal of iron impurity Amongst the given options, the compound(s) in which all the atoms are in one plane in all the possible conformations (if any), is (are) (A) H HC C-C H CH (B) H-CEC H -C CH (C) HC=C=O (D) HC=C=CH
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The density of a solution prepared by dissolving 120 g of urea (mol. mass = 60 u) in 1000 g of water is 1.15 g/mL. The molarity of this solution is (a) 2.05 M (b) 0.50 M (c) 1.78 M (d) 1.02 M
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
Find the magnitude of the reaction at A and the tension in cable CD using (a) the con- cepts of three-force bodies (closed polygon method discussed in chapter 2). (b) Check your answers by doing sum...
-
The periodogram in the Bartlett method can be expressed as Where r (i) xx (m) is the estimated autocorrelation sequence obtained from the i th block of data. Show that P (i) xx ( f ) can be expressed...
-
Consider the derivation of Equation (4.5) for the sample size of a two-sample z-test with known variance and equal allocation. a. Identify terms in Equation (4.2) that are smaller than $\alpha / 2$...
-
What is the procedure for substituting parties during the course of a pending lawsuit?
-
Frenchvanilla Company earned net income of $ 75,000 during the year ended December 31, 2014. On December 15, Frenchvanilla declared the annual cash dividend on its 5% preferred stock (par value, $...
-
Question 3 of 3 Additional information: 1. Net income for 2022 was $104,300. 2. Depreciation expense was $32,300. 3. Cash dividends of $46,300 were declared and paid. 4. Bonds payable with a carrying...
-
BCE has included service revenue of $22,100 as a result of a number of one year service policies sold late in December as an experiment. These service policies became effective on January 1, 20X4, or...
-
West Coast Hotel has 1 5 0 rooms. The occupancy rate varies between 5 0 % and 9 0 % per month, but the average occupancy rate is generally 8 0 % . In other words, on average, 8 0 % of the hotel s...
-
Identify the common pressure points (challenges) to expect at each stage (common SDLC stages). How to overcome these challenges? The common stages are: Initiation, Planning, Execution, Controlling...
-
Graph the polynomial function g(x)=-x(x-1)(x+4)
-
Suppose f"(x)=x-2 and the critical values of the function f'(x) are x= - Classify each critical value as either a relative minimum, relative maximum, or unknown using the second derivative test....
-
Consider the curve f(x) = (x + 1) + 4. Determine the points on the curve where the tangents to the curve pass through the point P(-2, 1). Show an algebraic solution and support your work with a graph.
-
Use the following information to prepare the current assets section of Lima Company's classified balance sheet as of December 31. Adjusted Trial Balance Cash and equivalents Accounts receivable...
-
Assume that Mr. Browns total farm asset is three times of his net worth and his total liability is $30,000. Calculate his total farm assets. b. Calculates Mr. Browns net worth. 2. Agland, Inc. has...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
The fuel in high-efficiency natural gas vehicles consists primarily of methane (CH4). (a) How much heat is produced in burning 1 mol of CH4(g) under standard conditions if reactants and products are...
-
Equation 21.28 is the nuclear reaction responsible for much of the helium-4 production in our Sun. How much energy is released in this reaction?
-
(a) What is meant by the term molecularity? (b) Why are termolecular elementary reactions so rare? (c) What is an intermediate in a mechanism?
-
A cylindrical tank of methanol has a mass of \(60 \mathrm{~kg}\) and a volume of \(75 \mathrm{~L}\). Determine the methanol's weight, density, and specific gravity. Take the gravitational...
-
A mass of \(0.5-\mathrm{kg}\) of argon is maintained at \(1400 \mathrm{kPa}\) and \(40^{\circ} \mathrm{C}\) in a tank. What is the volume of the tank?
-
The air in an automobile tire with a volume of \(0.015 \mathrm{~m}^{3}\) is at \(30^{\circ} \mathrm{C}\) and \(140 \mathrm{kPa}\) (gage). Determine the amount of air that must be added to raise the...

Study smarter with the SolutionInn App



