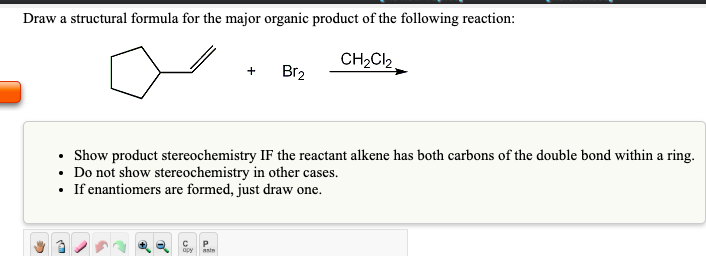
Draw a structural formula for the major organic product of the following reaction: Br CHCl, ...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
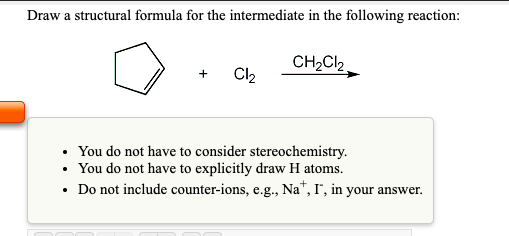
Draw a structural formula for the major organic product of the following reaction: Br₂ CH₂Cl₂, • Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. • If enantiomers are formed, just draw one. Draw a structural formula for the major organic product of the following reaction: CH₂Cl₂ + P opy aste Br₂ Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. If enantiomers are formed, just draw one. Draw a structural formula for the intermediate in the following reaction: Cl₂ . CH₂Cl₂ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. Do not include counter-ions, e.g., Na+, I", in your answer. Draw a structural formula for the major organic product of the following reaction: Br₂ CH₂Cl₂, • Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. • If enantiomers are formed, just draw one. Draw a structural formula for the major organic product of the following reaction: CH₂Cl₂ + P opy aste Br₂ Show product stereochemistry IF the reactant alkene has both carbons of the double bond within a ring. • Do not show stereochemistry in other cases. If enantiomers are formed, just draw one. Draw a structural formula for the intermediate in the following reaction: Cl₂ . CH₂Cl₂ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. Do not include counter-ions, e.g., Na+, I", in your answer.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Draw a structural formula for an optically active compound with the molecular formula a. C6H14 b. C5H11Br c. C4H10O d. C4H8Cl2
-
Draw the major organic product of the following reaction, and select the mechanism which would dominate (SN 1 , SN 2 , E 1 , or E 2 ). Draw all four bonds at chiral centers. Select the mechanism...
-
Draw the major organic product of the following reaction, and select the mechanism which would dominate (SN 1 , SN 2 , E 1 , or E 2 ). Draw all four bonds at chiral centers. Select the mechanism...
-
When Steve Jobs first demonstrated "the pinch"-the two-finger gesture to zoom in and out on photos and Web pages on the iPhone, it just rocked the mobile phone industry-the whole digital world heard...
-
Lager Dental Clinic is a medium-sized dental service specializing in family dental care. The clinic is currently preparing the master budget for the first 2 quarters of 2012. All that remains in this...
-
When the boss fired Clarence from his job at a moving company, she said it was because he could no longer lift heavy furniture; his salary was too high and, as he got older, he would have a hard time...
-
What qualitative factors should managers at Microbake consider with respect to their outsourcing decision?
-
The Regal Cycle Company manufactures three types of bicyclesa dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Management is concerned about the...
-
You are planning to make monthly deposits of $130 into a retirement account that pays 10 percent annual interest (APR), compounded monthly. If your first deposit will be made one month from now, how...
-
The adjusted trial balance of Gibson Company for the year ended December 31, 2019, is as follows: Instructions Prepare a multiple-step income statement, retained earnings statement, and a classified...
-
The Boy Manufacturing Co. manufactures a product which is processed in three successive departments - Department A, Department B, and Department C. In September, units were spoiled in all three...
-
The stock price of Nozama Inc. cannot be found online. According to forecasts, it will pay dividends that grow constantly. Similar stocks expect returns of 27.10%. According to Barren Wuffet, the...
-
A pitcher throws a 0.60 kg ball of clay at a 4.0 kg block of wood. The clay sticks to the wood on impact, and their joint velocity afterward is 2.5 m/s. What was the original speed of the clay (in...
-
Three years ago, you purchased a bond for $974.69. The bond had three years to maturity, a coupon rate of 8%, paid annually, and a face value of $1,000. Each year, you reinvested all coupon interest...
-
What do we know about the Galaxies over time according to https://webb.nasa.gov/content/science/galaxies.html What are some of the unknowns How might the JWT shed light on the galaxies over time?...
-
In Almond country, farmers are experimenting with new genetically modified franken-bees' that are highly effectively at pollinating their nut trees. However, the bees also produce a corrosive form of...
-
In Colorado last year, 8% of all new cars tested failed the state emmissions test. 20 new cars were selected at random to undergo an emmissions test. Answer the questions below
-
"Standard-cost procedures are particularly applicable to process-costing situations." Do you agree? Why?
-
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If the beverage is 0.265 M tartaric acid, what is the molal concentration? The density of the...
-
The equilibrium-constant expression for a gas reaction is Write the balanced chemical equation corresponding to this expression. Ke [C,Hg][O,]
-
Ethylene oxide, C2H4O, decomposes when heated to give methane and carbon monoxide. C2H4O(g) CH4(g) + CO(g) The following kinetic data were observed for the reaction at 688 K: Find the rate law and...
-
To estimate the optimal mix of debt and equity for your firm and to evaluate the effect on firm value of moving to that mix. Key Questions Based on the cost of capital approach, what is the optimal...
-
To determine whether your firm should move to its optimal mix (and if so, how) and to analyze the right type of debt for your firm. Key Questions If your firms actual debt ratio is different from...
-
HG Lang Designs operates an exclusive bridal boutique in Manhattan. All gowns are designed by Lang but are manufactured by various small firms in France and Italy. The following financial information...

Study smarter with the SolutionInn App


