Draw a structural formula of the S configuration of the compound shown below. a) b) c)...
Fantastic news! We've Found the answer you've been seeking!
Question:
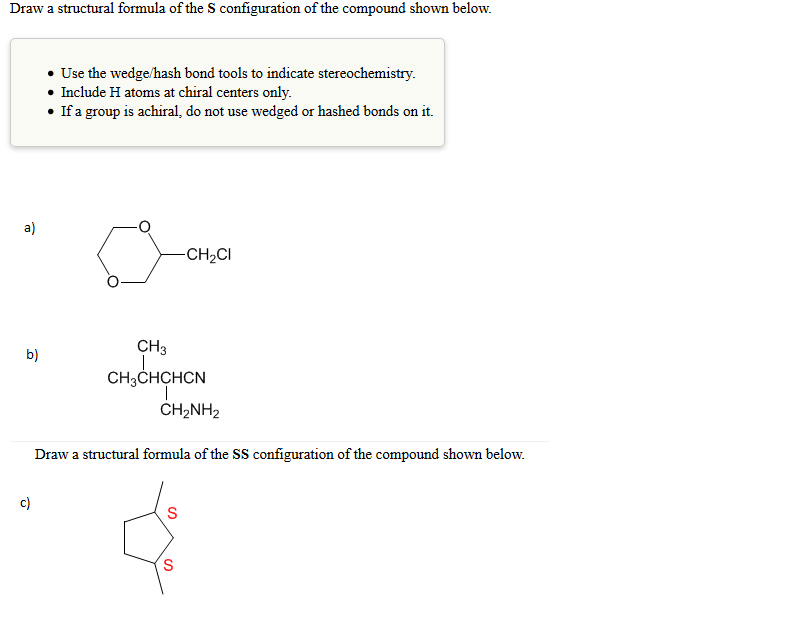
Transcribed Image Text:
Draw a structural formula of the S configuration of the compound shown below. a) b) c) • Use the wedge/hash bond tools to indicate stereochemistry. Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. CH3 CH3CHCHCN | -CH₂CI CH,NH2 Draw a structural formula of the SS configuration of the compound shown below. S S Draw a structural formula of the S configuration of the compound shown below. a) b) c) • Use the wedge/hash bond tools to indicate stereochemistry. Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. CH3 CH3CHCHCN | -CH₂CI CH,NH2 Draw a structural formula of the SS configuration of the compound shown below. S S
Expert Answer:
Answer rating: 100% (QA)
Answer Draw a structural formula of the S configuration of the compound shown below a b c Use ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Draw a structural formula for an optically active compound with the molecular formula a. C6H14 b. C5H11Br c. C4H10O d. C4H8Cl2
-
Draw a structural formula for each of the following. a. 3-methylpentanoic acid b. Ethyl methanoate c. Methyl benzoate d. 3-chloro-2,4-dimethylhexanoic acid
-
Draw a structural formula for each of the following compounds. (Several formulas may be possible in each case.) An alcohol with the molecular formula C5H10O
-
In Problems 65-72, summarize all pertinent information obtained by applying the graphing strategy, and sketch the graph of y=f(x). 66. 68. 70. 72. x2x6 f(x) 2x 1x +14 f(x)-_-x2-4 x3-5x2-6x 3x +2...
-
Is the following instrument a note, a check, or a draft? Why? If it is not a check, how would you have to change it to make it a check? To: Arthur Adams January 1, 2013 TEN DAYS AFTER DATE PAY TO THE...
-
1. Was the CNET story sufficient justification for the HP boards actions? Why or why not? 2. HP Chairwoman Patricia Dunn defended the actions of the board by arguing that HPs higher standards of...
-
Refer to the information from QS 21-18. Compute the variable overhead spending variance and the variable overhead efficiency variance and classify each as favorable or unfavorable. Data From QS 21-18...
-
Mallory Luongo, Inc. manufactures five models of kitchen appliances at its Mesa plant. The company is installing activity-based costing and has identified the following activities performed at its...
-
An ATV has an average acceleration of 1.9 m/s 2 . If the vehicle accelerates for 5.8 s and has an initial velocity of 15 m/s [E], what is the final velocity of the ATV?
-
Q1. Use the LIFO cost-flow assumption to answer the following questions. YEAR 1: Purchase #1 1,000 units @ $1 = $1,000 Purchase #2 1,000 units @ $1 = $1,000 a. How much is goods available for sale?...
-
The engineering department of a steel manufacturer is analyzing the companys rolling machine thats produces sheets of steel with varying thickness. The thickness is found to be a uniform random...
-
There are two charged q 1 = + 2 nC located at ( 2 , 3 ) and q 2 = - 3 nC located at ( 3 , 6 ) . Find the electrostatic force between the two
-
Sahil Inc. reported credit sales of $600,000 and cash collections of $450,000 for last year. The ending balance in accounts receivable was $800,000. Bad debt expense is estimated at 1% of credit...
-
Karina has a health insurance policy that includes a deductible of $500 and a coinsurance of 20 percent. If her total bill is $3,200, how much will she be required to pay?
-
52. A disk of mass 2.0 kg and radius 60 cm with a small mass of 0.05 kg attached at the edge is rotating at 2.0 rev/s. The small mass, while attached to the disk, slides gradually to the center of...
-
A factor costs $540,000. You forecast that it will produce cash inflows of $170,000 in year 1, $230,000 in year 2, & $400,000 in year 3. The discount rate is 11%. A) What is the value of the factory?...
-
Watertown, Inc. was founded in May, 2005 by Lawrence Rollins, who was employed for 20 years prior as a river rafting guide. Mr. Rollins received an unexpected inheritance of $2,000,000 in January,...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Give brief statements about the relevance of the following complexes in living systems: (a) Hemoglobin, (b) Chlorophylls, (c) Siderophores.
-
The oxidation of SO2 to SO3 is catalyzed by NO2. The reaction proceeds according to: NO2(g) + SO2(g) NO(g) + SO3(g) 2 NO(g) + O2(g) 2 NO2(g) (a) Show that the two reactions can be summed to give...
-
What observations made by Reinitzer on cholesteryl benzoate suggested that this substance possesses a liquid crystalline phase?
-
Role of controller, role of chief financial officer. George Jimenez is the controller at Balkin Electronics, a manufacturer of devices for the computer industry. The company may promote him to chief...
-
In the following, assume that all growth and discount rates are stated in real terms. 1. Assume the Eurozone inflation-adjusted average growth in capital stock is 3.0 percent per annum into...
-
Which conclusion presented by Ryan about the top-down approach and the bottom-up approach is most likely correct? A. Conclusion 1. B. Conclusion 2. C. Conclusion 3. Use the following information to...

Study smarter with the SolutionInn App


