Draw the Lewis structure of SF4 showing all lone pairs. What is the hybridization of the...
Fantastic news! We've Found the answer you've been seeking!
Question:

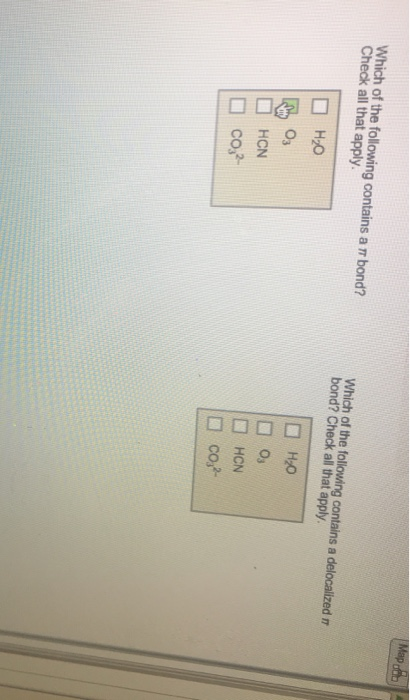
Transcribed Image Text:
Draw the Lewis structure of SF4 showing all lone pairs. What is the hybridization of the central atom? sp sp³d sp³d An SF4 molecule is polar. nonpolar. Identify the molecular geometry of SF4. square pyramidal linear bent tetrahedral octahedral see-saw T-shaped trigonal pyramidal square planar trigonal planar trigonal bipyramidal What are the ideal bond angles of this geometry? Choose all that apply. 90 degrees 109.5 degrees 120 degrees 180 degrees yious Give Up & View Solulion Check Answer Next Exit Which of the following contains a r bond? Check all that apply. H₂O 03 HCN co²- Which of the following contains a delocalized n bond? Check all that apply. H₂O 0₁ HCN co²- Map 000 Draw the Lewis structure of SF4 showing all lone pairs. What is the hybridization of the central atom? sp sp³d sp³d An SF4 molecule is polar. nonpolar. Identify the molecular geometry of SF4. square pyramidal linear bent tetrahedral octahedral see-saw T-shaped trigonal pyramidal square planar trigonal planar trigonal bipyramidal What are the ideal bond angles of this geometry? Choose all that apply. 90 degrees 109.5 degrees 120 degrees 180 degrees yious Give Up & View Solulion Check Answer Next Exit Which of the following contains a r bond? Check all that apply. H₂O 03 HCN co²- Which of the following contains a delocalized n bond? Check all that apply. H₂O 0₁ HCN co²- Map 000
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the hybridization of the central atom in (a) SiCl4 (b) HCN (c) SO3 (d) TeCl2.
-
Draw the Lewis structure of O2F2. Assign oxidation states and formal charges to the atoms in O2F2. The com-pound O2F2 is a vigorous and potent oxidizing and fluo-rinating agent. Are oxidation states...
-
Draw the lewis structure and identify the three molecular geometry for CH3SO2H, C(CH2)2 and CH3NCO
-
Below is the comparative income statement of Variline, Inc. Requirements 1. Prepare a horizontal analysis of the comparative income statement of Variline, Inc. Round percentage changes to the nearest...
-
Pretzelmania, Inc., issues 7%, 15-year bonds with a face amount of $70,000 for $64,008 on January 1, 2015. The market interest rate for bonds of similar risk and maturity is 8%. Interest is paid...
-
What is a package in UML approach?
-
Harold Fail owned a plot of land in Georgia known as Ebenezer Landing, where he operated a public boat ramp and parking lot. In 1999, Fail and Steve Bodiford entered into an oral agreement...
-
The following material relates to Darrow Company: Required Place an X in the appropriate columns for each of thesituations. Cash Flows Classification Noncash Effect on Cash Operating Investing...
-
Figure 1 shows a flowchart of nested if. Produce a java programming code segment of the flowchart. Start False True True Fine=0.00 Speed-110 Speed 120 Fine - 120 True Figure 1 False True Speed <130...
-
Blossom Corporation is preparing the comparative financial statements to be included in the annual report to stockholders. Blossom employs a fiscal year ending May 31. Income from operations before...
-
The 5-kg slender bar is released from rest in the horizontal position shown. At the instant when it has rotated 45, its angular velocity is 4.16 rad/s. At that instant, you need to determine the...
-
Review | Constants | Periodic Table A 4.5 g latex balloon is filled with 2.6 g of helium. When filled, the balloon is a 31-cm-diameter sphere. When released, the balloon accelerates upward until it...
-
Use this article below to give the best example with supporting points to make a example of an Annotated biography ...
-
Your company is considering launching a new line of stoves. The manufacturing plant required for producing the new line of stoves costs $40,000,000 (today) and will be depreciated down to zero over...
-
X transfers prop (basis $5K/ FMV $60K) for 600 shares of a newly formed Aces Corp. Y receives 400 shares in Aces corp. for services rendered to the corp. Each share is worth $100. Do X and/or Y have...
-
Pls code according to the description. And pls tidy your code - a snippet would be really appreciated and i will rate helpful right away for a correct answer. Lastly DONT GOOGLE or FAKE the answer i...
-
Calculate the productivity, SLOC per work month, of each of the projects in the following table, and also for the organization as a whole. If the project leaders for projects "a" and "d" had...
-
Catherine (aged 42) and Johnson (aged 45) have been married for 12 years. Johnson is a project manager of an event company at a monthly salary of $55,000 with an additional one-month salary of...
-
The heat of fusion of water is 6.008 10 3 J mol 1 at its normal melting point of 273.15 K. Calculate the freezing point depression constant K f .
-
Use the logic diagram of Figure 27.2 to determine the point group for PCl 5 . Indicate your decision-making process as was done in the text for NH 3 . a. linear? b. C n axis? c. more than 1C n axis?...
-
Consider the Daniell cell, for which the overall cell reaction is Zn(s) + Cu 2+ (aq) Zn 2+ (aq) + Cu(s). The concentrations of CuSO 4 and ZnSO 4 are 2.50 10 3 and 1.10 10 3 m, respectively. a....
-
Figure 1.10 shows an automatic water level control system. (a) Explain how the automatic control system works indicating the roles of key components. (b) If the automatic water level control system...
-
Many closed-loop and open-loop control systems can be found in homes. (a) List six such examples (three open-loop and three closed-loop). (b) Construct feedback control system block diagrams for the...
-
The diagram depicted in Figure 1.38 is a closed-loop temperature control system. (a) Explain how this control system works. (b) If the automatic controller is replaced by manual control, explain how...

Study smarter with the SolutionInn App


