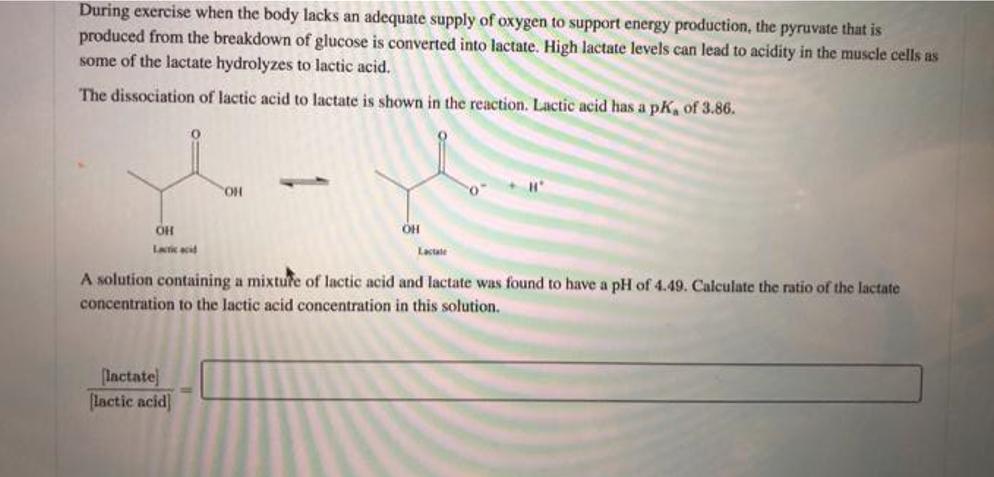
During exercise when the body lacks an adequate supply of oxygen to support energy production, the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
During exercise when the body lacks an adequate supply of oxygen to support energy production, the pyruvate that is produced from the breakdown of glucose is converted into lactate. High lactate levels can lead to acidity in the muscle cells as some of the lactate hydrolyzes to lactic acid. The dissociation of lactic acid to lactate is shown in the reaction. Lactic acid has a pK, of 3.86. OH Lactic acid OH [lactate] [lactic acid] Lactate H" A solution containing a mixture of lactic acid and lactate was found to have a pH of 4.49. Calculate the ratio of the lactate concentration to the lactic acid concentration in this solution. During exercise when the body lacks an adequate supply of oxygen to support energy production, the pyruvate that is produced from the breakdown of glucose is converted into lactate. High lactate levels can lead to acidity in the muscle cells as some of the lactate hydrolyzes to lactic acid. The dissociation of lactic acid to lactate is shown in the reaction. Lactic acid has a pK, of 3.86. OH Lactic acid OH [lactate] [lactic acid] Lactate H" A solution containing a mixture of lactic acid and lactate was found to have a pH of 4.49. Calculate the ratio of the lactate concentration to the lactic acid concentration in this solution.
Expert Answer:
Answer rating: 100% (QA)
The detailed answer for the above question is provided below HendersonHasselb... View the full answer

Related Book For 

Biochemistry Concepts and Connections
ISBN: 978-0321839923
1st edition
Authors: Dean R. Appling, Spencer J. Anthony Cahill, Christopher K. Mathews
Posted Date:
Students also viewed these chemistry questions
-
The breakdown of glucose to pyruvate utilizes the free energy of 2 ATP molecules; however, the synthesis of glucose from pyruvate requires 4 ATP. Using the basic ideas of thermodynamics presented in...
-
The pyruvate ( lactate reaction in animals is reversible, but the pyruvate ( ethanol fermentation in yeast is irreversible. Explain.
-
When a person exercises, muscle contractions produce lactic acid. Moderate increases in lactic acid can be handled by the blood buffers without decreasing the pH of blood. However, excessive amounts...
-
Return to Better Mousetraps in Problem 18. Suppose the firm can cut its requirements for working capital in half by using better inventory control systems. By how much will this increase project NPV?...
-
An E-R diagram can be viewed as a graph. What do the following mean in terms of the structure of an enterprise schema? a. The graph is disconnected. b. The graph is acyclic.
-
Eason Company manufactures wheel rims. The controller expects the following ABC allocation rates for 2018: Eason produces two wheel rim models: standard and deluxe. Expected data for 2018 are as...
-
Calculate backlog depreciation at 31 December 2005 for the plant and equipment, the details of which are given in Question 30.5A. Data From Question 30.5A:- The plant and equipment, details of which...
-
On January 1, 2008, Michelle Co. issued ten-year bonds with a face value of $1,000,000 and a stated interest rate of 10%, payable semiannually on June 30 and December 31. The bonds were sold to yield...
-
The selection and choose the best answer Not to Keep by Robert Frost They sent him back to her. The letter came Saying... And she could have him. And before She could be sure there was no hidden ill...
-
P12-11A The comparative balance sheets for Spicer Company as of December 31 are presented below. SPICER COMPANY Comparative Balance Sheets December 31 Assets 2017 2016 $ 68,000 50,000 151,450 15,280...
-
Bronco Company uses normal costing in its job-costing system. The company produces custom bikes for toddlers. The beginning balances (December 1) and ending balances (as of December 30) in their...
-
Write down the characteristics of an ideal solution.
-
Mention the usefulness of the van Laar equation to determine the activity coefficients of a binary solution.
-
Derive Margules two-suffix three-parameter equation for the calculation of activity coefficients.
-
Differentiate between UNIFAC and UNIQUAC methods.
-
In case of flash distillation, how do you calculate the flash?
-
Take the stock price of RIL BSE. Assuming a strike price of 1800. a) What is the 30-day VaR? b) Establish the price of 3-month European Call Option using Monte-Carlo Simulation c)?...
-
In a certain school district, 3% of the faculty use none of their sick days in a school year. Find the probability that 5 faculty members selected at random used no sick days in a given year.
-
Assume a pH gradient of 4.0 units across a thylakoid membrane, with the lumen more acidic than the stroma. (a) What is the standard free energy change per mol of protons associated with this gradient...
-
Each gram of mammalian skeletal muscle consumes ATP at a rate of about 1 Ã 10-3 mol/min during contraction. To bridge the short interval between the moderate demand for ATP met by aerobic...
-
Consider the following questions about glutamate dehydrogenase. (a) The reaction as shown on page 569 has NH3 as a reactant, instead of NH4+, which is far more abundant at physiological pH. Why is...
-
Why do overhead costs often shift from high-volume products to low-volume products when a company switches from a conventional costing method to activity-based costing?
-
The Grange Company had the following results for the year to 31 March 19X1. A single product - a toggle - was made by the company. The standard cost of manufacturing each unit was \( 1.50\). What are...
-
A You are required to calculate the overhead variances of Raymond Ltd. The budget is prepared as: (a) Total budgeted variable overhead \( 100,000\). (b) Total budgeted fixed overhead \( 125,000\)....

Study smarter with the SolutionInn App


